Introduction
Bladder cancer is the tenth most commonly diagnosed cancer
worldwide [1]. The age-standardized incidence rate (per 100,000
person-years) is 9.5 for men and 2.4 for women worldwide [1].
Smoking is the most important risk factor for bladder cancer,
accounting for slightly less than 50% of cases [2]. Although the
majority of patients present with non-muscle-invasive disease,
approximately 30-40% of patients already have MIBC at the time
of initial diagnosis due to the insignificance of early clinical symptoms, with a poor prognosis [3]. The traditional treatments for
MIBC mainly include radical cystectomy (RC) and chemotherapy,
but there is a high recurrence rate, and the 5-year overall survival
(OS) rate remains at 15–20% [4]. Nearly 50% of MIBC patients
who underwent radical bladder resection still developed metastatic bladder cancer [5]. However, neoadjuvant chemotherapy
(NAC) can reduce the risk of death in patients with MIBC after RC
and improve the OS [6,7]. At present, it has become the standard
treatment recommended by multiple MIBC guidelines [8].
Although RC has historically been the cornerstone of treatment
for MIBC, optimizing outcomes with NAC and alternative options
for bladder preservation strategies have also emerged as treatment options [8,9]. The choice of treatment regimen for MIBC patients is directly related to the final prognosis, and preoperative
NAC can create opportunities for subsequent tumor resection.
There are many NAC schemes for MIBC, among which gemcitabine combined with cisplatin (GC) and methotrexate, vincristine,
doxorubicin combined with cisplatin (MVAC) are the two most
widely used schemes in clinical application [8]. Preoperative NAC
for MIBC can help control local lesions, reduce tumor stage, reduce surgical difficulty, eliminate micro-metastases, and improve
post-operative long-term survival rate. Compared with RC alone,
NAC and RC resulted in a 16% increase in the 5-year OS rate [10].
As the utilization of NAC for MIBC has increased and post-NAC
ypT0 rates have remained steady at 35–40%, many have questioned the need for RC at all [11]. In fact, a recent study concluded
that while patients who achieved pathological complete response
(pCR) lived longer, patients who did not achieve pCR had a worse
prognosis than those who underwent direct RC without NAC [12].
As a result, insensitivity to NAC in some patients leads to tumor
progression, overtreatment, and delayed surgery. Therefore, it is
an urgent and difficult problem to predict the sensitivity of NAC
for bladder cancer [13]. Many tentative efforts have been made
to solve this problem. Soichiro Yoshida et al. investigated whether
Diffusion-weighted MRI (DW-MRI) could predict NAC sensitivity
of MIBC and concluded DW-MRI is a potential biomarker [14]. But
the sample size was too small to draw a wide conclusion. Recent
evidence suggests that bladder urothelial cancers harboring mutations in DNA damage response (DDR) pathways are associated
with improved pathologic responses to NAC [15] and prolonged
survival [16]. Contrary to this, Russell E.N.Baker et al. argued that
clinical restaging and tumor sequencing are inaccurate indicators
of response to NAC for MIBC [11]. Thus, predicting the sensitivity
of NAC for bladder cancer patients has not been solved yet, which
is a clinical problem to be solved urgently.
In the current study, we sought to understand whether the
clinical and pathologic tools currently available in clinical practice
are sufficient to determine individual patient responses to NAC
accurately and reliably in a retrospective population. We downloaded the clinical data and the immunohistochemical results of
149 bladder cancer patients from the attachment of the paper
published by Gottfrid Sjodahl et al. [17]. Multiple machine learning algorithms were used to screen and identify some genes and
clinical features that were differentially expressed in different
pathological reactions. Finally, the prediction model of NAC therapy for bladder cancer was constructed according to the 10 factors
included. Our study identified some genes closely associated with
NAC in bladder cancer and constructed predictive models for the
efficacy of NAC. Furthermore, this study also provides ideas and
clues to bladder cancer therapy, and the identified genes could
also be considered as NAC biomarkers for bladder cancer.
Materials and methods
Data source and Pre-processing
We downloaded the clinical data of 149 bladder cancer patients and the immunohistochemical results of some genes from
the attachments of the paper published by Gottfrid Sjodahl et al
[17], and the data used in this study is attached in supplementary
data 1. According to the pathological response of NAC, patients
were divided into pCR, pathological partial response (pPR) and
no pathological response (no pR) groups, and the clinical baseline
data of patients in different groups were statistically analyzed and
sorted out. The independent T test was used for measurement
data and the Chi-square test was used for enumeration data.
Among them, the clinical characteristics with P<0.1 were incorporated into the factors of subsequent model construction. Then, R
software (R Foundation for Statistical Computing, Vienna, Austria)
was used for subsequent analyses.
Influence of NAC pathological response on prognosis
There is a close relationship between the efficacy of NAC and
the prognosis of bladder cancer. To clarify the relationship between pathological response of NAC and prognosis of bladder
cancer, the Kaplan-Meier (K-M) analysis for OS, cancer-specific
survival (CCS) and recurrence free survival (RFS) were proceeded
based on the NAC pathological response groups with the aid of R
software and the Log-Rank was utilized to test. The survival curve
was plotted using the survminer R package.
Identification of genes associated with NAC efficacy
We compared the expression of the 16 genes detected by immunohistochemistry (IHC) among the three groups via the Krus-kal-Wallis test to explore genes closely related to NAC. Finally,
genes with P<0.1 were selected for mapping and demonstration
using ggplot2 R package.
Survival analysis for genes related to NAC
To clarify the effect of genes associated with NAC efficacy on
prognosis, six genes with P<0.1 were selected for K-M survival
analysis. During the process, the K-M analysis for OS were proceeded based on the IHC expression of genes whose cut-off level
was set at the median value with the aid of R software and the
Log-Rank was utilized to test.
Lasso regression and correlation analysis
To prevent the over-fitting of the prediction model and further
screen the clinical factors and genes expression, we selected the significant factors for Lasso regression analysis, and analyzed the
correlation of IHC expression levels of the selected genes. Lasso
regression was completed using glmnet R package, and the correlation analysis was completed by using corrplot R package. The selected factors were used for the next step of model construction.
Predictive model construction and comparison
The significant factors screened via Lasso regression was used
for model construction to predict the pathological response of
NAC in patients with bladder cancer. Model construction methods
include random forest (RF), support vector machine (SVM) and
neural network (NN). Among which, RF analysis was completed
via randomForest R package, SVM analysis via e1071 R package,
and NN analysis via neuralnet R package. All 149 samples were
used for the training set, and 30% of them were randomly selected as internal validation for the model validation. Then, we
compared the accuracy, sensitivity, specificity and the area under
curve (AUC) of the three different prediction models. Finally, we
select the NN model with better prediction performance, and
demonstrate the importance of screening factors and the prediction accuracy of the NN model.
Results
Clinical baseline data
We compared the clinical characteristics of three different
pathological response groups to NAC, and the detailed results
were shown in (Table 1). Among them, age, T, N, TURB path LVI,
keratinization, CIS, Necrosis, RB_alt_status, and P16_alt_status
are strongly correlated with NAC efficacy. These variables were
used for further screening and analysis in subsequent Lasso regression.
Influence of NAC pathological reaction on prognosis
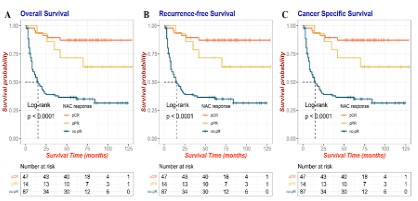
Survival analysis and comparison among the no pR, pPR and
pCR groups showed that the prognosis of patients in the pCR and
pPR groups was much better than that of patients with no pR for
OS (Figure 1A), RFS (Figure 1B) and CSS (Figure 1C). This also indicates that the pathological response of NAC plays a very important role in the prognosis of bladder cancer patients, so predicting
the efficacy of NAC therapy is also an urgent problem to be solved.
Table 1: Clinical baseline data.
|
no pR |
pCR |
pPR |
P |
| Total |
87 |
48 |
14 |
|
| Age (years) |
|
|
|
0.125 |
| <60 |
20 |
6 |
5 |
|
| ≥60 |
67 |
42 |
9 |
|
| Sex (Male) |
68 |
37 |
11 |
0.987 |
| T stage |
|
|
|
0.0008 |
| T1 |
0 |
1 |
0 |
|
| T2 |
24 |
31 |
4 |
|
| T3 |
49 |
15 |
8 |
|
| T4 |
14 |
1 |
2 |
|
| N stage |
|
|
|
0.254 |
| N0 |
75 |
42 |
12 |
|
| N1 |
9 |
1 |
1 |
|
| N2 |
3 |
3 |
1 |
|
| N3 |
0 |
2 |
0 |
|
| M |
|
|
|
0.347 |
| M0 |
87 |
47 |
14 |
|
| M1 |
0 |
1 |
0 |
|
| TURB path LVI (yes) |
37 |
16 |
1 |
0.03 |
| TURB path keratinization (yes) |
25 |
7 |
1 |
0.051 |
| TURB path CIS (yes) |
19 |
10 |
6 |
0.201 |
| TURB path necrosis (yes) |
56 |
28 |
5 |
0.094 |
| Histologic Variant (yes) |
16 |
7 |
1 |
0.535 |
| RB_alt_status (mutation) |
30 |
24 |
4 |
0.147 |
| P16_alt_status (mutation) |
44 |
13 |
7 |
0.026 |
| LundTax IHC subtype |
|
|
|
0.388 |
| Ba/Sq |
15 |
3 |
0 |
|
| GU |
27 |
19 |
5 |
|
| Mes-like |
2 |
1 |
0 |
|
| NE-like |
5 |
4 |
0 |
|
| Uro |
37 |
21 |
9 |
|
| Consensus.subtype (%) |
|
|
|
0.779 |
| BASQ |
23 |
8 |
2 |
|
| LumNS |
10 |
4 |
2 |
|
| LumP |
20 |
16 |
5 |
|
| LumU |
13 |
10 |
2 |
|
| NE_like |
7 |
2 |
0 |
|
| StromaRich |
14 |
8 |
3 |
|
| NAC protocol |
|
|
|
0.818 |
| GC |
51 |
24 |
7 |
|
| MVAC |
34 |
23 |
7 |
|
| Other |
2 |
1 |
0 |
|
| NAC courses |
|
|
|
0.815 |
| 2 |
5 |
1 |
0 |
|
| 3 |
57 |
34 |
10 |
|
| 4 |
16 |
10 |
4 |
|
| 5 |
4 |
1 |
0 |
|
| 6 |
5 |
2 |
0 |
|
Identification of genes associated with NAC efficacy
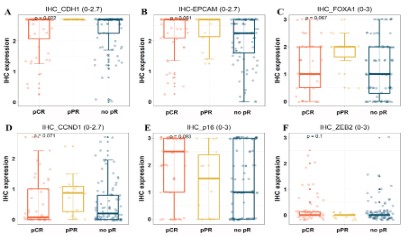
The IHC expression values of 16 genes were analyzed and compared among different pathological response groups, and the
six genes with P<0.1 were selected and showed in (Figure 2A-F).
These six genes are CDH1, EPCAM, FOXA1, CCND1, P16 and ZEB2,
respectively. These genes are associated with NAC response in
bladder cancer, and can be used to predict NAC response. Moreover, these genes are also potential NAC therapy targets for bladder cancer.
Survival analysis for six genes related to NAC
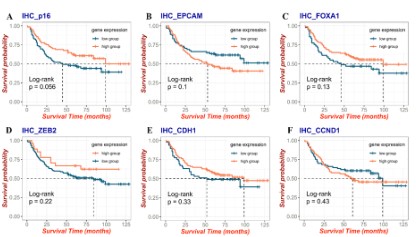
K-M analysis for OS was used to explore the prognostic value
of the six genes associated with NAC and the results were shown
in (Figure 3). We found that although these six genes were not
significant differences with prognosis, there were obvious differences between high and low expression groups. Therefore, these
six genes are related to prognosis, and were used for further
screening and analysis in subsequent Lasso regression.
Lasso regression and correlation analysis
The results of Lasso regression (Figure 4A-B) showed that 10
of the 15 NAC-related factors can be used for model construction, while the other 5 factors may have potential collinearity,
which would affect the fitting degree of the model. The correlation analysis of the 6 NAC-related genes is shown in Figure 4C, and
we could find that CCND1 was significantly correlated with FOXA1 and P16, while CDH1 was significantly correlated with FOXA1 and
ZEB2. Therefore, CCND1 and CDH1 were excluded by Lasso regression analysis. Figure 4D shows the importance ranking of the
10 genes included in lasso regression.
Predictive model construction and comparison
The 10 clinical characteristics or genes expression were used
to construct the prediction model. The main analysis results of
RF/SVM/NN models are shown in supplementary data 2, and
the comparison results of the three models are shown in (Table
2). Among the three models, the NN model has higher accuracy
(93.8%) and AUC (0.984). It can predict the efficacy of NAC for
bladder cancer more accurately.
Table 2: Comparison of prediction results of three prediction
models in train set.
|
Correct percentage |
AUC |
| Train set |
overall |
no pR |
pCR |
pPR |
| NN |
93.8% |
96.3% |
97.9% |
64.3% |
0.984 |
| RF |
81.3% |
79.3% |
83.3% |
85.7% |
na |
| SVM |
89.6% |
95.1% |
85.4% |
71.4% |
0.948 |
Demonstration of prediction results of NN model
The NN model has the best prediction accuracy, so we present
the main results of the NN model. (Figure 5A) shows the importance of various predictive factors in the NN model, among which
age and IHC_FOXA1 were of higher importance. (Figure 5B) shows
ROC curves of different pathological reactions, and the AUC value
of no pR, pPR and pCR were 0.985, 0.974 and 0.996, respectively.
(Figure 5C) shows the confusion matrix of training set and verification set. It can be seen that the NN model has high accuracy in
both training set and internal validation set.
Discussion
Currently, the clinical guidelines of the European Association
of Urology and the American Society of Clinical Oncology have
recommended NAC for the preoperative treatment of MIBC [18-20]. Nonetheless, patients may be ineligible for either chemotherapy [21] or RC, and selection of the most appropriate therapy
depends on a staging system that may not accurately determine
the eligibility of a patient for a specific treatment plan [22]. Thus,
a more personalized approach to bladder NAC therapy management is warranted [13].
The results of our study showed that the prognosis of bladder
cancer patients whose NAC efficacy reached pCR was much better than that of patients without pathological reaction, which was
consistent with previous studies [6,7]. However, for patients with
no pR, NAC therapy may cause delay of operation time, delay of
surgery and overtreatment, resulting in the progression and poor
prognosis of bladder cancer. Therefore, in this study, available
clinical features and pathological data were used to explore the
genes and clinical features closely related to NAC for bladder cancer, and to construct a prediction model for the efficacy of NAC for
bladder cancer patients.
The clinical factors included in this study are age, T stage, N
stage, TURB path LVI (lymphatic invasion) and keratinization. Age
is one of the most important risk factors for cancer, and the occurrence and prognosis of cancer patients is highly influenced by
age and ageing [19,23]. Studies have shown that older patients
are less likely to receive NAC [24], and this may be one reason for
the poor prognosis in older patients with bladder cancer. Clinical stages are associated with the indications of NAC therapy and
RC in bladder cancer patients [9]. NAC with cisplatin-based combinations has been standard for decades in patients with resectable N0M0-invasive bladder cancer [18]. Moreover, LN-positive patients previously treated with NAC have a poor prognosis, sig-
nificantly worse than LN-positive patients subsequently treated
with AC, and should be considered for protocols using sandwich
chemotherapy approaches or novel agents [25]. Therefore, NAC
therapy should not be used in patients with positive lymph nodes.
Intracavitary keratosis of bladder cancer was also one of the factors associated with NAC in this study. Hendrik Jütte et.al [20] retrospectively analyzed the association of basal and luminal mRNA
expression patterns such as Keratin 20 (KRT 20), Keratin 5 (KRT5),
as well as ESR1 and ERBB2 in patients with MIBC at transurethral
resection (TUR-BT) with pCR at RC after platinum-based NAC, and
found that tumors with elevated expression of markers associated
with intracavity differentiation (KRT20, ERBB2, ESR1) was associated with a higher incidence of pCR. Therefore, all the clinical factors included in this study have a certain relationship with bladder
cancer NAC.
In this study, the four genes screened into the prediction model are P16, EPCAM, ZEB2 and FOXA1. These genes are closely associated with NAC efficacy and prognosis of bladder cancer. P16
is a tumor suppressor gene directly involved in cell cycle regulation, and deletion or mutation of P16 is very common in tumors
[26,27]. In bladder cancer, the expression of P16 is significantly
reduced, and it is associated with various molecular subtypes of
bladder cancer [28]. The molecular subtypes of bladder cancer
are closely related to the efficacy of NAC therapy [17]. EPCAM
expression is increased in bladder cancer and is associated with
lymph node metastasis [29]. EPCAM is mainly involved in bladder
cancer metastasis and chemotherapy resistance through PI3K/
AKT signaling pathway [30]. Moreover, antibody-drug conjugations targeting EPCAM represent a novel therapeutic approach
for urothelial carcinoma [31]. Expression of ZEB2 correlates with
histopathological grade in papillary urothelial tumors of the urinary bladder [32]. In addition, ZEB2 is involved in the invasion and
metastasis of bladder cancer [33]. FOXA1 is expressed throughout
the urothelium, and associated with the urothelial differentiation
process and the molecular subtypes of bladder cancer. There is
a near relationship between MIBC tumor subtypes (identified by
IHC) and the response to NAC and survival [12]. In brief, P16, EPCAM, ZEB2 and FOXA1 are well correlated with NAC efficacy and
prognosis of bladder cancer.
In this study, three different machine learning algorithms were
used to construct prediction models for the efficacy of NAC for
bladder cancer. Among them, the neural network prediction model has high accuracy. The advantage of this study lies in the use
of a variety of clinical characteristics and genes expression tested
by IHC to predict the efficacy of NAC. Secondly, the prediction
model is highly reliable by comparing various methods. However,
this study also has shortcomings. The sample size of this study
is small, and the model is only verified internally, but not externally. A larger sample size and more scientific research methods
are needed to predict the efficacy of NAC for bladder cancer in
the future.
Conclusion
In this study, available clinical features and gene expression detected by IHC were used to explore factors closely related to NAC
for bladder cancer, and to construct prediction models to predict
the efficacy of NAC for bladder cancer patients. This study not only finds several targets for NAC and provides reference for the
feasibility of NAC therapeutic prediction model for bladder cancer, but also provides a new clue for other researchers and clinical
workers to predict the efficacy of NAC.
Declarations
Conflict of interest: All the authors declare no conflicts of interest.
Funding: This work was supported by the Open Foundation of
Gansu Key Laboratory of Functional Genomics and Molecular Diagnostics and Gansu Province Intellectual Property Planning project (21ZSCQ012).
Data availability: All data and material analyzed can be obtained from the corresponding author.
References
- Organization WH, Cancer IAfRo. Estimated number of new cases in
2018, worldwide, both sexes, all ages. In: 2018.
- van Osch FH, Jochems SH, van Schooten FJ, Bryan RT, Zeegers MP.
Quantified relations between exposure to tobacco smoking and
bladder cancer risk: a meta-analysis of 89 observational studies.
International journal of epidemiology. 2016; 45(3): 857-870.
- Lewis GD, Haque W, Verma V, Butler EB, Teh BS. The Role of Adjuvant Radiation Therapy in Locally Advanced Bladder Cancer. Bladder cancer (Amsterdam, Netherlands). 2018; 4(2): 205-213.
- Aggen DH, Drake CG. Biomarkers for immunotherapy in bladder
cancer: a moving target. Journal for immunotherapy of cancer.
2017; 5(1): 94.
- Stenzl A, Cowan NC, De Santis M, et al. The updated EAU guidelines on muscle-invasive and metastatic bladder cancer. European
urology. 2009; 55(4): 815-825.
- Petrelli F, Coinu A, Cabiddu M, Ghilardi M, Vavassori I, Barni S.
Correlation of pathologic complete response with survival after
neoadjuvant chemotherapy in bladder cancer treated with cystectomy: a meta-analysis. European urology. 2014; 65(2): 350-357.
- Sherif A, Holmberg L, Rintala E, et al. Neoadjuvant cisplatinum
based combination chemotherapy in patients with invasive bladder cancer: a combined analysis of two Nordic studies. European
urology. 2004; 45(3): 297-303.
- Cathomas R, Lorch A, Bruins HM, et al. The 2021 Updated European Association of Urology Guidelines on Metastatic Urothelial
Carcinoma. European urology. 2022; 81(1): 95-103.
- Aragon-Ching JB, Werntz RP, Zietman AL, Steinberg GD. Multidisciplinary Management of Muscle-Invasive Bladder Cancer: Current
Challenges and Future Directions. American Society of Clinical Oncology educational book American Society of Clinical Oncology Annual Meeting. 2018; 38: 307-318.
- Anan G, Hatakeyama S, Fujita N, et al. Trends in neoadjuvant chemotherapy use and oncological outcomes for muscle-invasive
bladder cancer in Japan: a multicenter study. Oncotarget. 2017;
8(49): 86130-86142.
- Becker REN, Meyer AR, Brant A, et al. Clinical Restaging and Tumor
Sequencing are Inaccurate Indicators of Response to Neoadjuvant
Chemotherapy for Muscle-invasive Bladder Cancer. European urology. 2021; 79(3): 364-371.
- Bhindi B, Frank I, Mason RJ, et al. Oncologic Outcomes for Patients
with Residual Cancer at Cystectomy Following Neoadjuvant Chemotherapy: A Pathologic Stage-matched Analysis. European urology. 2017; 72(5): 660-664.
- Shah JB, McConkey DJ, Dinney CP. New strategies in muscle-invasive bladder cancer: on the road to personalized medicine. Clinical
cancer research : an official journal of the American Association for
Cancer Research. 2011; 17(9): 2608-2612.
- Yoshida S, Koga F, Kobayashi S, et al. Role of diffusion-weighted
magnetic resonance imaging in predicting sensitivity to chemoradiotherapy in muscle-invasive bladder cancer. International journal of radiation oncology, biology, physics. 2012; 83(1): e21-27.
- Plimack ER, Dunbrack RL, Brennan TA, et al. Defects in DNA Repair Genes Predict Response to Neoadjuvant Cisplatin-based Chemotherapy in Muscle-invasive Bladder Cancer. European urology.
2015; 68(6): 959-967.
- Teo MY, Bambury RM, Zabor EC, et al. DNA Damage Response and
Repair Gene Alterations Are Associated with Improved Survival in
Patients with Platinum-Treated Advanced Urothelial Carcinoma.
Clinical cancer research : an official journal of the American Association for Cancer Research. 2017; 23(14): 3610-3618.
- Sjödahl G, Abrahamsson J, Holmsten K, et al. Different Responses
to Neoadjuvant Chemotherapy in Urothelial Carcinoma Molecular
Subtypes. European urology. 2021.
- Witjes JA, Bruins HM, Cathomas R, et al. European Association of
Urology Guidelines on Muscle-invasive and Metastatic Bladder
Cancer: Summary of the 2020 Guidelines. European urology. 2021;
79(1): 82-104.
- Chang SS, Bochner BH, Chou R, et al. Treatment of Non-Metastatic
Muscle-Invasive Bladder Cancer: AUA/ASCO/ASTRO/SUO Guideline. The Journal of urology. 2017; 198(3): 552-559.
- Jütte H, Reike M, Wirtz RM, et al. KRT20, KRT5, ESR1 and ERBB2 Expression Can Predict Pathologic Outcome in Patients Undergoing
Neoadjuvant Chemotherapy and Radical Cystectomy for Muscle-Invasive Bladder Cancer. Journal of personalized medicine. 2021;
11(6).
- Galsky MD, Hahn NM, Rosenberg J, et al. A consensus definition
of patients with metastatic urothelial carcinoma who are unfit for
cisplatin-based chemotherapy. The Lancet Oncology. 2011; 12(3):
211-214.
- Compérat EM, Burger M, Gontero P, et al. Grading of Urothelial
Carcinoma and The New “World Health Organisation Classification
of Tumours of the Urinary System and Male Genital Organs 2016”.
European urology focus. 2019; 5(3): 457-466.
- Kendal WS. Dying with cancer: the influence of age, comorbidity,
and cancer site. Cancer. 2008; 112(6): 1354-1362.
- Gan T, Chen Q, Huerta CT, Huang B, Evers BM, Patel JA. Neoadjuvant Therapy in Stage II/III Rectal Cancer: A Retrospective Study in
a Disparate Population and the Effect on Survival. Diseases of the
colon and rectum. 2021; 64(10): 1212-1221.
- Cha EK, Sfakianos JP, Sukhu R, Yee AM, Sjoberg DD, Bochner BH.
Poor prognosis of bladder cancer patients with occult lymph node
metastases treated with neoadjuvant chemotherapy. BJU international. 2018; 122(4): 627-632.
- Dizdar O, Demirci U. p53 status in locally advanced bladder cancer.
Journal of clinical oncology : official journal of the American Society of Clinical Oncology. 2012; 30(3): 339; author reply 339-340.
- Carneiro FP, Amorim RF, de Vasconcelos Carneiro M, et al. P16 as
a marker of carcinoma in effusions and peritoneal washing. BMC
cancer. 2020; 20(1): 225.
- Olkhov-Mitsel E, Hodgson A, Liu SK, Vesprini D, Xu B, Downes MR.
Three-antibody classifier for muscle invasive urothelial carcinoma
and its correlation with p53 expression. Journal of clinical pathology. 2021.
- van der Fels CAM, Rosati S, de Jong IJ. EpCAM Expression in Lymph
Node Metastases of Urothelial Cell Carcinoma of the Bladder: A Pilot Study. International journal of molecular sciences. 2017; 18(8).
- Zuo Y, Xu X, Chen M, Qi L. The oncogenic role of the cerebral endothelial cell adhesion molecule (CERCAM) in bladder cancer cells in
vitro and in vivo. Cancer medicine. 2021; 10(13): 4437-4450.
- Lattanzi M, Rosenberg JE. The emerging role of antibody-drug
conjugates in urothelial carcinoma. Expert review of anticancer
therapy. 2020; 20(7): 551-561
- Lee H, Jun SY, Lee YS, Lee HJ, Lee WS, Park CS. Expression of miRNAs and ZEB1 and ZEB2 correlates with histopathological grade in
papillary urothelial tumors of the urinary bladder. Virchows Archiv:
an international journal of pathology. 2014; 464(2): 213-220.
- Sun DK, Wang JM, Zhang P, Wang YQ. MicroRNA-138 Regulates
Metastatic Potential of Bladder Cancer Through ZEB2. Cellular
physiology and biochemistry : international journal of experimental cellular physiology, biochemistry, and pharmacology. 2015;
37(6): 2366-2374.