Introduction
Surgeons learn to perform complex manual skills through systematic practice [1-3]. Skillful performance is characterized by increased movement speed and accuracy of [4-6], likely involving
the transition from goal-to habit-directed behavioral control and
the development of movement automation [7,8].
In humans, brain activity during complex movements has been
assessed primarily by EEG recordings of simple self-paced movements and visually- and auditory-triggered movements. Simple
voluntary movements modulate the alpha rhythm (8-12 Hz) in the
visual and somatosensory-motor areas. Specifically, hand movements cause an alpha decrease or desynchronization, also known
as mu rythm, in the hand areas and a synchronization in the foot
areas [9,10]. The degree of synchronization also varies with the
phase of movement, showing a decrease prior to movement followed by an increase during movement [9,11,12]. These results
show an alpha modulation in the somatosensory-motor cortices
in opposite directions in different phases of movement, which
should result in little or no modulation of mean alpha amplitude
for continuous movements. Conversely, hand and foot voluntary
movements increase alpha amplitude over posterior electrodes
[11,12] resulting in a synchronization of the alpha band during
movement execution [13] and movement observation [14]. Summarizing, alpha amplitude in the occipital electrodes increases
during the execution of continuous movements.
The two main hypothesis about the mechanism of the alpha
rhythm are the absence of sensory input and the inhibition of
task-irrelevant networks. The first named the ‘idle rhythm hypothesis’ [15] posits that alpha synchronization is a consequence of a
reduced bottom-up sensory input [9,15]. The second hypothesis
posits that alpha synchronization is a consequence of a bottom-down inhibition of task irrelevant areas [16-18], together with a
redirection of the processing resources towards areas relevant
to the task [19]. In addition, alpha oscillations have been associated with the selection of attended objects and the inhibition
of non-attended distractors [20], in the visual and tactile modalities. A decrease in alpha has been observed over parieto-occipital
areas cotralateral to the attended visual hemifield [21,22] and
an increase over the occipital areas contralateral to the ignored
stimuli [22] for visual stimuli. Similarly, there is an increase in occipital alpha amplitude correlated with performance at the posterior electrodes [23], indicating a general modulation in a tactile
task. For the alpha inhibition theory, optimal performance on a
task should correlate with an increase in alpha amplitude in areas
where attentional resources are not needed to perform the task
[20]. Thus, alpha oscillations in the occipital area for a complex
visuo-sensorimotor task are probably the result of a redistribution of the attentional resources away from the occipital areas,
which predominate in the resting state, evidenced as an increase
in alpha amplitude over the occipital areas.
Alpha rhythm modulation by task demands was shown in posterior electrodes in visual and auditory attentional tasks [24-27]
and in central posterior and bilateral areas with greater visual
working memory load [24,28]. Although still controversial, the
evidence suggests an effect of task demand in the amplitude of
the alpha band. Surgical suturing can occur under self-paced conditions for simple sutures without time constraints or under fast-paced conditions, when there are time constraints and interfer-
ences that impose additional task demands. To the best or our
knowledge, there is no evidence on the modulation of the alpha
band with task-demands in complex visuo-sensorimotor tasks.
In addition, task expertise might modulate alpha amplitude
in task-related areas. Changes in relative regional activation of
brain areas have been observed in skill learning where practice
and subsequent consolidation leads to reorganization of the functional architecture of the brain [26]. Expert drivers have smaller
volume recruitment of task-related regions compared to novices
[27]. For surgical suture, early stages of learning are characterized
by prefrontal cortical activation, which attenuate with deliberate
practice [26] and movements automation [7], in agreement with
a modulation of the executive and attentional resources. For complex movements, behavioral measures of automaticity may disrupt the continuous flow of movements. An alternative approach
for a correlate of automaticity is the Electroencephalogram (EEG)
activity in the alpha-band [28,29]. In a laparoscopic surgical simulation, a continuous recording showed greater alpha power in
the occipital area in efficient or fast- compared to non-efficient or
slow-performers [30]. In summary, the evidence from surgeons
suggests that greater performance might correlate with greater
amplitude of alpha in posterior areas for complex movements.
In conclusion, complex visuo-sensorimotor tasks, such as suturing, involve the redistribution of the attentional resources away
from the visual areas predominantly engaged in resting eyes-open
condition, which might be modulated by expertise, task demand
and/or movement efficiency. This redistribution should materialize in a greater synchronization in the alpha band in the posterior
areas. We recorded scalp EEG of expert and novice surgeons to
evaluate whether the alpha band (8-12 Hz) in the occipital area is
modulated during continuous suture performance relative to resting and if this modulation is further shaped by surgical expertise,
task demand and/or movement efficiency. We obtained the power of alpha in the occipital area and the number of sutures of surgeons and medical students while performing continuous open
suture in both self-paced and fast-paced conditions and compared
it with the resting condition. We hypothesized that alpha power
should show (i) an effect of movement performance revealed by
a greater alpha power in both surgeons and medical students in
posterior areas for suture execution relative to resting; (ii) an effect of expertise revealed by a greater alpha power in surgeons
relative to medical students, (ii) an effect of task demand revealed
by a greater alpha power in both surgeons and medical students
with high task demands and (iv) an effect of suture performance
revealed by a positive association between occipital alpha activity
and suture performance during suturing.
Methods
Participants
Thirty one participants with normal or corrected-to-normal vision participated in this study. Nineteen were medical students (8
women and 11 men, mean age of 23.1) and twelve were surgeons
(5 women and 7 men, mean age of 49.5), all right-handed. All participants were recruited by invitation and gave written informed
consent before the recording session.
The procedures were conducted following the Protocol #46-
2020, approved by the Ethics Committee (Comité Ético Científico)
of the Universidad de Talca, in accordance with The Code of Ethics
of the World Medical Association (Declaration of Helsinki).
Procedure
To evaluate the amplitude of the alpha oscillations in experts
and novices surgeons, we performed EEG measurements while
participants performed suture in both self-paced and fast-paced
conditions as well as in resting condition. Novices were fourth-year medical students with 1 to 2 hours of standardized training in
a suture workshop in the School of Medicine, University of Talca.
Experts were established physicians with a minimum of 3 years
and a maximum of 20 years with regular suture procedures prac-ticing surgeons.
In a quiet room, participants were seated in front of a small
table containing the surgery pad, tools and suture (Figure 1A).
All participants were instructed on how to perform the sutures
in a simulation model 3/0 of 75 cm (Braun a video the surgical
technique) in a wound closure pad (Jig Mk 3 skin pad, Limbs and
things, GA, USA), standard surgical instruments and 75 cm nylon
3/0 sutures (Braun Hessen, Germany) before recordings.
Scalp EEG recordings were obtained in 2 conditions: resting
and open suture. In the resting condition, participants were quiet
for 3 min with their eyes open. In the suture condition, participants performed 6 suturing trials of 5 minutes each (Figure 1B),
divided into 3 trials of self-paced and 3 of fast-paced suturing with
an inter-trial interval of 2 minutes. The sequence of the self-paced
and fast-paced trials was randomized and balanced across participants. In self-paced suturing, participants were instructed to
perform the sutures at their own pace and in fast-paced suturing, participants were instructed to perform the greatest number of sutures and received a comment every 60 sec indicating
to increase the sutures because the other participates had better
results.
EEG recordings
The electroencephalogram (EEG) was continuously recorded
while participants completed all conditions, using a 32-channel
BioSemi ActiveTwo system (BioSemi B.V., Amsterdam, Netherlands): scalp sites (Fp1, Fp2, F7, F3, Fz, F4, F8, T7, C3, Cz, C4, T8,
P9, P7, P5, P3, P1, Pz, P2, P4, P6, P8, P10, PO7, PO3, POz, PO4,
PO8, O1, Oz, O2, and Iz) according to the modified 10–20 System
(American Electroencephalographic Society, 1994) plus 5 additional electrodes: left and right mastoids and 3 electro-oculogram
(EOG) channels (at outer canthi of each eye, and below the right
eye). All signals were recorded in single-ended mode. The EEG
and EOG were low-pass filtered with a 5th-order sync filter (half-
power cutoff at 208 Hz) and digitized at 1024 Hz.
Data analysis
Data analyses were conducted using a combination of EEGLAB
[31] and ERPLAB [32], running on MATLAB 2015a (MathWorks,
Natick, MA, USA). EEG signals were bandpass filtered offline using a non-causal Butterworth infinite impulse response filter, with
half-power cutoffs at 0.1 and 40 Hz, and a roll-off of 12 dB/octave,
and then down-sampled to 256 Hz. Eye-movement artifacts and
eye blinks were corrected using Independent Component Analysis (ICA). Subsequently, scalp channels were referenced offline to the
average of the left and right mastoids, and the three EOG signals,
plus Fp2, were used to create two new bipolar vertical and horizontal EOG derivations, to explore remaining ocular artifacts.
For each EEG recording, the first 4 seconds of data were removed from all trials to minimize the presence of artifacts. After that, data segments of 2.5 and 4.5 minutes were extracted
from resting and suture trials, respectively. The data from trials
for suture conditions were merged for each condition resulting
in 13.5 minutes of data. EEG data was subjected to a Fast Fourier
Transform with a 4-sec, 50% overlap, Hanning-taper, artifact-free
moving window. Power spectra with a number of averaged windows of less than 180 were eliminated from further analysis. The
grand average power spectra (μV2) were computed for each EEG
channel for all recordings. Thus, based on the scalp distribution of
alpha power (8-12 Hz) in occipital area (Figure 3), a ROI of three
occipital electrodes (O1, O2 and Oz) was defined, and obtained a
mean alpha for these electrodes. We additionally, evaluated the
mean alpha power in a ROI for the central electrodes (C3, Cz, C4).
Finally, mean power in the alpha band, from each participant’s
ROI, were compared across conditions. In addition, we obtained
the number of sutures (stitches) from each participant, for each
trial at each condition. The data from this study is available in
http://dx.doi.org/10.17632/xb8fzmrf8j.1.
Statistical analysis
Statistical differences were estimated by Bayesian analysis
using the Bayes Factor Toolbox for Matlab (https://github.com/
klabhub/bayesFactor in Matlab written by Bart Krekelberg) based
on Rouder et al. 2012 [33]. Statistical differences in power values were evaluated by a two-factor analysis of variance (ANOVA),
with between-subjects factor of expertise (two levels: expert
and novice) and within-subject factor of condition (three levels:
resting, self-paced and fast-paced). Statistical differences between the numbers of sutures were evaluated using a two factor
ANOVA. Main effects for the ANOVA were estimated as the ratio
of the Bayes factors for the full model and the restricted model,
obtained by excluding the factor from the full model. Differences
between means were assessed as the Bayes factor (BF) for paired
or unpaired t-tests. Bayes Factor for the H1 hypothesis equal or
greater than 10 (H1 > 0.90) indicates a strong evidence for the
alternative hypothesis and a BF for the H1 between 3 and 10 was
considered as moderate evidence for the alternative hypothesis.
To evaluate the association between alpha power and the efficiency of suture movements, we computed the Bayes factor for
the Pearson product-moment correlation coefficient. A strong
correlation was defined when r2 values were equal or greater than
0.5, and a moderate correlation when r2 values were between
0.45 and 0.5. Unless otherwise specified, all values are reported
as mean + SD in the main text and as mean + SEM in the figures.
Results
The main goal of this work was to evaluate if the execution of
complex movements modulates the amplitude in the alpha band
relative to resting, and if this modulation was further shaped by
expertise, task demand and suture performance. Alpha power
and suture performance were compared in expert and novice
surgeons in self-paced and fast-paced suturing and in resting www.journalonsurgery.org 4
conditions as an indicator of the redistribution of the inhibitory-attentional resources away from the occipital areas. From a total
of 31 participants, 12 surgeons and 19 medical students, 2 from
the medical student group were discarded due to the high noise
in the EEG recordings. Consequently, we report the results from
12 surgeons and 17 medical students.
Suture performance
To evaluate the efficiency of suture movements, we obtained
the mean number of sutures for each participant at each of three
5-minute trials in the self- and fast-paced conditions, (Figure 1).
As expected, surgeons had approximately twice the mean
number of sutures than medical students in both self-paced (M
= 10.2, SD = 2.1 vs M = 4.6, SD = 1.5, respectively) and fast-paced
(M = 12.1, SD = 1.5 vs M = 5.5, SD = 1.1, respectively) conditions
(Figure 2).
Moreover, the number of sutures was greater in fast-paced relative to self-paced condition surgeons and medical students. The
2 x 2 repeated-measures ANOVA on the number of sutures with
between-subject factor of expertise (surgeons, medical students)
and within-subject factor of task demand (self-paced, fast-paced)
show main effects of expertise (F(1, 54) = 18.2, BF = 3.4* 1010)
and task demand (F(1, 54) = 17.4, BF = 134.6), and no interaction between expertise and task demand (F(1, 54) = 3.2 , BF =
0.53). These results demonstrate that surgical expertise increases
suturing performance and that higher task demands add an extra
boost to performance. Post-hoc tests confirm that the number of
sutures was greater in surgeons (BF = 1.6* 1016, probability from
data for the alternative hypothesis, PH1 > .99, unpaired t-test)
than in medical students when data was pooled across task demands and that a greater demand in the fast-paced condition increased the number of sutures (BF = 80.5, PH1 > .98, paired t-test)
when data from surgeons and medical students were pooled.
Moreover, greater demand increases the number of sutures by
20 % in surgeons (BF = 8.1, PH1 = .89, paired t-test), but our data
does not support an effect of task demand in performance (BF =
2.8, PH1 > .74, paired t-test) for medical students. In summary,
these results demonstrate that surgical expertise almost doubles
the speed of suture movements and that a greater task demand
increases the speed of movements in surgeons.
Modulation of the alpha rhythm
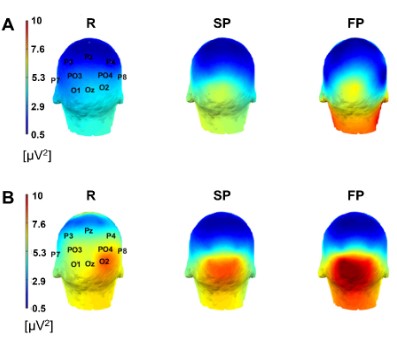
The average scalp maps of the power in the alpha band for surgeons and medical students, plotted with ERPLAB [32] / EEGLAB
[31], are shown in Figure 3. In surgeons, a baseline alpha power
in posterior electrodes O1, O2 and Oz was observed in resting (R),
eyes open condition. The alpha power in the posterior electrodes
increased during suture movement execution in self-paced (SP)
and fast-paced (FP) conditions (Figure 3A). A similar pattern was
obtained in medical students (Figure 3B).
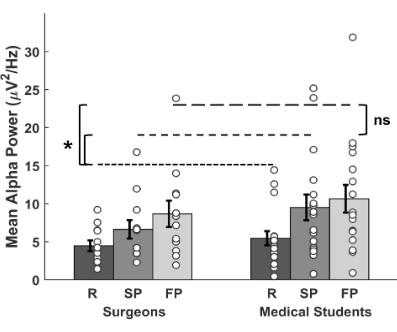
To quantify the variation in the alpha power in open suture,
a ROI in the occipital area (O1, Oz and O2, see methods section)
was defined based on the scalp distribution. The mean alpha
power for each ROI for resting, self-paced and fast-paced suturing
is shown in Figure 4. In surgeons, alpha power was higher during suturing for both self-paced and fast-paced conditions (M =
6.63, SD = 4.21, and M = 8.66, SD = 6.02, respectively) relative to
resting (M = 4.47, SD = 2.47, Figure 4). Likewise, alpha power was higher in medical students when suturing in self-paced and fast-paced conditions (M = 9.50, SD = 6.96, and M = 10.64, SD = 7.49
s, respectively) relative to resting (M = 5.45, SD = 3.83, Figure 4).
The 2 x 3 ANOVA for the individual alpha power values with between-factor of expertise (surgeons, medical students) and with-in-factor of task type (resting, self-paced, fast-paced) showed a
main effect of task type (F(1, 83) = 8.0, BF = 1.34
* 10
3), no effect of
expertise (F(1, 83) = 0.54, BF = 0.26), and no interaction between
expertise and task type (F(1, 83) = 0.26, BF = 0.08). These results
indicate that suturing increases the power in the alpha band in
the occipital electrodes relative to resting and that alpha power
was not additionally modulated by expertise and task demand.
Post-hoc tests show a greater alpha power in self-paced (BF = 9.4,
PH1 = .90, paired t-test) and fast-paced (BF = 145.0, PH1 > .99)
suturing relative to resting, but no difference in alpha power between self-paced and fast-paced suturing (BF = 1.7, PH1 = .63),
after collapsing for expertise. In summary, our results show that
switching from resting to open suturing increases the amplitude
of the alpha band in the occipital electrodes, in agreement with
a redistribution of the attentional resources away from occipital
areas during suture execution.
Moreover, because alpha activity is modulated by hand movements in sensorimotor cortex [9,11,12], we evaluated the alpha
power in a ROI for the central electrodes (C3, Cz, C4). In surgeons,
the mean alpha power was M = 1.49, SD = 0.90, M = 1.44, SD =
0.81, and M = 2.07, SD = 2.59, in resting, self-paced and fast-paced
conditions and in medical students the alpha power was M = 3.11,
SD = 5.41 M = 1.46, SD = 0.91, and M = 1.60, SD = 1.09, in resting, self-paced and fast-paced conditions. The ANOVA indicated
no main effect of expertise (F(1, 83) = 3.26, BF = 0.01) and task
type (F(1, 83) = 0.68, BF = 0.07), and no interaction between these
factors (F(1, 83) = 4.25, BF = 0.64). Summarizing, the mean alpha
power in the sensorimotor areas was not modulated in continuous suturing.
Association between the alpha rhythm and suture performance
We show that surgical expertise increases the efficiency of
surgical movements and that the execution of surgical suture increases the strength of alpha activity in the occipital areas in both
surgeons and medical students. Besides, suturing efficiency was
strongly modulated by task demand in surgeons but not in medical students, which could indicate that expert surgeons may have
a greater control of movement efficiency. In parallel, there as a
trend for greater alpha power over occipital areas with increasing task demand, suggesting an association between movement
efficiency and alpha power in the occipital areas as observed in
laparoscopic movements [10]. To test for a relationship between
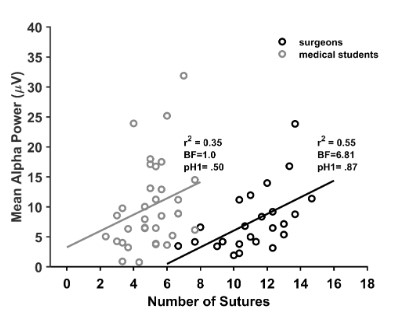
the alpha amplitude and suture efficiency, we assessed the correlation of the occipital alpha power during suturing with suture efficiency in surgeons and medical students. The scatterplot of the
individual alpha power values in surgeons and medical students in
relaxed and high-demand conditions as a function the number of
sutures is shown in Figure 5.
However, our results do not support the modulation of mean
alpha amplitude by expertise and task demand.
The Pearson correlation coefficient show a linear positive correlation between the individual alpha power and the number of
sutures for surgeons (r2(22) = 0.55), with a moderate statistical evidence from the data (BF = 6.81, pH1 = .87), suggesting that as the
suture efficiency increases, there is a greater power in the alpha
band. In contrast, our data does not support the correlation in
medical students (r2(32) = 0.35, BF = 1.0, pH1 = .50). These results
suggest that greater movement performance during suturing associates with increases in occipital alpha power for expert surgeons.
Discussion
We evaluated whether alpha amplitude in the posterior areas is modulated by movement execution, expertise and task
demands in a continuous complex visuo-sensorimotor task. To
do so, we obtained the number of sutures, as a measure of the
efficiency of movements and the mean alpha power, as an estimate of the redistribution of attentional resources, in self-paced
or low-demand suturing and fast-paced or high-demand suturing
and resting eyes open condition in expert and novice surgeons.
As expected, we show that surgical expertise markedly increases the total number of sutures indicating a greater movement efficiency in both self-paced and fast-paced suturing and that greater task demand in fast-paced suturing further enhances
the total number of sutures in expert surgeons, but not significantly in medical students. Second, we show an increase in the
amplitude of the alpha band in the posterior electrodes during suture performance relative to resting in both, surgeons and medical students, in agreement with a task associated redistribution of
the attentional resources. In addition, expertise and task demand
do not further modulate alpha power, indicating no additional effect on the redistribution of attentional resources. Finally, alpha
amplitude correlated positively with the number of sutures only
in surgeons, suggesting a linear dependency between the redistribution of the attentional resources and movement efficiency. In
the following paragraphs we address each of the findings.
Regarding open suture, surgical expertise nearly doubled the
number of sutures, a behavioral measure of movement efficiency,
relative to medical students in both self-paced and fast-paced
conditions (Figure 2), in agreement with previous evidence of a
greater precision and lower variability of expert motor execution
in expert surgeons [2-5,34-36]. The difference in performance
between novices and experts likely reflect the difference from
goal-directed and habit-directed movements, respectively, which
involve distinct patterns of cortical and subcortical activation
[7,8,37]. Task demand further modulates the number of sutures
by 20 % in surgeons (Figure 2) in response to the instruction to
achieve superior number of sutures as well as the interference
in the form of comparative comments with other participants,
indicating that experts can additionally speed up movements. In
contrast, our data does not support an effect of task demand in
medical students, even with a larger sample size (N = 17), presumably due to greater variability in movement performance in
low and high demand conditions. In conclusion, the long-term effect of expertise improves suture performance by 100 % and the
short-term effect of task-demand improves performance by an
additional 20% in experts.
Regarding the power in the alpha band, execution of a continuous complex visuo-sensorimotor task results in an overall
alpha synchronization (Figure 4) in the occipital electrodes (Figure 3) in both, surgeons and medical students, relative to resting.
These results are consistent with previous studies showing synchronization in the posterior electrodes for simple finger or foot
movements [11,12] and movement observation [14]. Similar effects were observed in motor [38], somatosensory [39], auditory
[40,41], visual attention [21,22] and internal/external attention
tasks [42]. The greater alpha amplitude is not consistent with full
inhibition of “task irrelevant” occipital areas because visual input
is required for suture movements. Alternatively, our results sug-
gest a redistribution of limited attentional resources throughout
cortical areas when switching tasks from resting to open suture,
as observed when closing the eyes in complete darkness [42] or
when directing attention internally [24,25], in agreement with the
attentional theory of the alpha rhythm [17,18].
In contrast, expertise does not modulate the mean alpha power (Figure 3), suggesting that the difference in motor control between novices and experts does not impact the redistribution of
attentional resources away from occipital areas. Imaging of motor
skill acquisition has shown substantial changes in brain activation
between the initial and later stages of learning, indicating a gradual transition from goal- to habit-like movements [8,37]. Medical students in the early stages of skill learning, probably perform
open sutures trough goal-like movements entailing a high activation of the anterior areas involved in attentional control and
executive function [43-47]. Meanwhile, expert surgeons probably
perform sutures trough habit-like movements with greater activation of the sensory areas and basal ganglia and lower activation
of the frontal cortex [8,37,38]. In conclusion, our results suggest
that although different expertise levels in open suture involve
substantial differences in brain processing, these differences are
not expressed over the mean alpha power in the posterior occipital areas.
Likewise, task demand elicited no additional modulation of
the mean alpha band (Figure 4), suggesting that the processes
engaged in the redistribution of attentional resources over the
posterior electrodes were not shaped by task demand in agreement with previous results [25]. In conclusion, task demand did
not modulate the overall alpha band in the occipital areas.
In addition to the effect of movement execution on the alpha
power, our results suggest a modulation by movement efficiency
in expert surgeons, in agreement with the general increase in alpha power in posterior electrodes in a somatosensory discrimination task [23]. Our data from 12 surgeons show a positive linear
association between alpha power in the posterior areas and the
number of sutures. Although with a moderate statistical support
from the data, these results suggest that the extent of experts’
attentional redistribution away from the occipital areas in open
suture is associated with movement efficiency, in agreement with
a greater alpha power in the occipital regions in good performers obtained in laparoscopic surgery [30]. Moreover, imaging of
movement speed revealed the activation of multiple cortical and
subcortical areas, with slow movements involving the prefrontal
areas bilaterally and fast movements involving the sensorimotor cerebral cortex [49,50], excluding the posterior occipital areas. Additional studies with a greater sample size should be performed to confirm this association. In contrast, our data does not
support this association in medical students, probably because
of the different processes involved in movement execution and because there is no significantly higher movement efficiency for
fast-paced condition despite the larger sample size.
Because simple movements trigger increases and decreases
of alpha synchronization in the somatosensory and motor areas
within a movement cycle [9,11,13,15], we also evaluated alpha
activity over the central electrodes (C3, Cz and C4) in the somatosensory areas during suture execution. We found no difference
in the mean alpha amplitude for suture execution, and no effect
of expertise and task demand, as anticipated for a continuous
recording of alpha power in the suturing trials where amplitude
modulations should cancel-out throughout the suturing exercise.
While the results presented here contribute to the understanding of the modulation of the alpha rhythm in the occipital areas
in a more ecological setting, consisting in a continuous execution
of complex movements and its relationship with movement performance, they also have several limitations. First, we obtained
the mean alpha power in the occipital areas during continuous
suture movements not stereotyped across time and participants,
hindering the identification EEG signals associates to specific
movements’ phases. The failure to associate the time course of
the alpha power for complex movements is clearly a limitation of
this study, particularly for the central electrodes located over the
somatosensory and motor control areas of the hand [13].
Future studies should evaluate the time course of alpha amplitude in different cortical areas for specific movement phases.
Second, our study had a small sample size of expert surgeons (N
= 12), despite the efforts set in the recruitment. Several limitations of small sample sizes include an overestimation of the effect
size and more type II errors [52]. To reduce the effect of a limited
sample size, we performed Bayesian statistics whose outcomes
(Bayes Factors) indicate the support of the data for the null and alternative hypothesis. Because of the small sample limitation, further studies should be done to corroborate the modulation of the
alpha amplitude in open suture. Finally, the mean age of surgeons
and medical students was different, but we found no statistical
difference in the mean alpha power for resting, indicating that the
age difference of surgeons and medical students was not a source
of variability for the alpha power for resting baseline condition.
Conclusions
In summary, here we provide evidence for the synchronization
in the alpha band in the occipital areas in open suture, both in
surgeons and in medical students relative to the resting and its
association with the efficiency of movements. Surgeons are twice
as efficient in the number of open sutures, likely due to movement automation. High task-demand increases suture efficiency
by 20% only in surgeons, in agreement with additional movement
speed enhancement in expert surgeons. Interestingly, the overall
mean amplitude of EEG alpha band is greater for suturing relative to resting, although it is not modulated by expertise and task-demand, in agreement with a redistribution of the attentional
resources away from occipital areas during movement execution.
Surgeons have a positive linear association between the occipital alpha amplitude and suture efficiency, suggesting that visuo-sensorimotor expertise may reshape the relationship between
movement execution and alpha-related networks. In contrast,
medical students have no association between alpha amplitude
and movement efficiency, likely based in a greater complexity in the control of suture movement. Taken together, these findings
are consistent with the redistribution of limited-resources held by
the inhibitory-attentional theories of alpha rhythm in a complex
visuo-sensorimotor task.
Declarations
Conflicts of interest: The authors declare that they have no
conflicts of interest.
Funding sources: This work was supported by Convenio U. de
Talca - U. de Chile (#1557) and Programa de Investigación (PIA)
en Ciencias Cognitivas, Centro de Investigación en Ciencias Cognitivas (CICC), Facultad de Psicología, Universidad de Talca, Chile.
Authors' contributions: SR designed the experiment, collected and analyzed the data, edited the article; MQ collected and
analyzed the data; JK analyzed the data and wrote the article; JLC
analyzed the data and wrote the article; MLA designed the experiment, analyzed the data and wrote the article. All authors read
and approved the final manuscript.
Ethics approval and consent to participate: The procedures
were conducted with Protocol #46-2020 in accordance with
The Code of Ethics of the World Medical Association (Declaration of Helsinki). The study was approved by Scientific Ethic
Committee (Comité Ético Científico) of the Universidad de Talca.
All participants were recruited by invitation and gave written informed consent before the recording session.
Availability of data and materials: The datasets supporting the
conclusions of this article are available in the Mendeley reposi-
tory, Reyes, Sergio; Quinones, Matias; Kreither, Johanna; Lopez-
Calderon, Javier; Aylwin, Maria (2021), “Alpha Waves Open Su-
ture”, Mendeley Data, V1, DOI: 10.17632/xb8fzmrf8j.1
Abbreviations: EEG: Electroencephalogram; EOG: Electro-Ocu-
logram; R: Resting; SP: Self-Paced; FP: Fast-Paced; ROI: Region of
Interest; BF: Bayes Factor; ANOVA: Analysis of Variance; SD: Stan-
dard Deviation; SEM: Standard Error of the Mean
References
- Ching SS, Mok CW, Koh YX, Tan SM, Tan YK. Assessment of surgical
trainees’ quality of knot-tying. J Surg Educ. 2013; 70: 48-54.
- Mashaud LB, Arain NA, Hogg DC, Scott DJ. Development, validation, and implementation of a cost-effective intermediate-level
proficiency-based knot-tying and suturing curriculum for surgery
residents. J Surg Educ. 2013; 70: 193-199.
- Huang E, Chern H, O’Sullivan P, Cook B, McDonald E, et al. A better way to teach knot tying: a randomized controlled trial comparing the kinesthetic and traditional methods. Am J Surg. 2014; 208:
690-694.
- Gölz C, Voelcker-Rehage C, Mora K, Reuter EM, Godde B, et al.
Improved Neural Control of Movements Manifests in Expertise-Related Differences in Force Output and Brain Network Dynamics.
Front Physiol. 2018; 9: 1540.
- Hofstad EF, Våpenstad C, Chmarra MK, Langø T, Kuhry E, et al. A
study of psychomotor skills in minimally invasive surgery: what differentiates expert and nonexpert performance. Surg Endosc 2013;
27: 854-863.
- Horeman T, Blikkendaal MD, Feng D, van Dijke A, Jansen F, et al. Visual force feedback improves knot-tying security. J Surg Educ.
2014; 71: 133-141
- Ashby FG, Turner BO, Horvitz JC Cortical and basal ganglia contributions to habit learning and automaticity. Trends Cogn Sci. 2010;
14: 208-215.
- Bouton ME Context, attention, and the switch between habit and
goal-direction in behavior. Learn Behav. 2021; 49: 349-362.
- Pfurtscheller G, Stancák A, Jr., Neuper C Event-related synchronization (ERS) in the alpha band--an electrophysiological correlate of
cortical idling: a review. Int J Psychophysiol. 1996: 24; 39-46.
- Pfurtscheller G, Neuper C Event-related synchronization of mu
rhythm in the EEG over the cortical hand area in man. Neurosci
Lett. 1994; 174: 93-96
- Pfurtscheller G, Lopes da Silva FH Event-related EEG/MEG synchronization and desynchronization: basic principles. Clin Neurophysiol. 1999; 110: 1842-1857.
- Pfurtscheller G Induced oscillations in the alpha band: functional
meaning. Epilepsia 2003: 44: 2-8.
- Neuper C, Pfurtscheller G Event-related dynamics of cortical
rhythms: frequency-specific features and functional correlates. Int
J Psychophysiol. 2001; 43: 41-58.
- Di Nota PM, Chartrand JM, Levkov GR, Montefusco-Siegmund R,
DeSouza JFX Experience-dependent modulation of alpha and beta
during action observation and motor imagery. BMC Neurosci.
2017; 18: 28.
- Adrian ED, Matthews BHC The Berger rhythm: potential changes
from the occipital lobes in man. Brain. 1934; 57: 355-385.
- Suffczynski P, Kalitzin S, Pfurtscheller G, Lopes da Silva FH Computational model of thalamo-cortical networks: dynamical control of
alpha rhythms in relation to focal attention. Int J Psychophysiol.
2001; 43: 25-40.
- Klimesch W, Sauseng P, Hanslmayr S. EEG alpha oscillations: the
inhibition-timing hypothesis. Brain Res Rev. 2007; 53: 63-88.
- Jensen O, Mazaheri A. Shaping functional architecture by oscillatory alpha activity: gating by inhibition. Front Hum Neurosci. 2010;
4: 186
- Sadaghiani S, Scheeringa R, Lehongre K, Morillon B, Giraud AL, et
al. Intrinsic connectivity networks, alpha oscillations, and tonic
alertness: a simultaneous electroencephalography/functional
magnetic resonance imaging study. J Neurosci. 2010; 30: 10243-
10250.
- Klimesch W α-band oscillations, attention, and controlled access
to stored information. Trends Cogn Sci. 2012; 16: 606-617.
- Sauseng P, Klimesch W, Stadler W, Schabus M, Doppelmayr M, et
al. A shift of visual spatial attention is selectively associated with
human EEG alpha activity. Eur J Neurosci 2005; 22: 2917-2926.
- Worden MS, Foxe JJ, Wang N, Simpson GV Anticipatory biasing of
visuospatial attention indexed by retinotopically specific alpha-band electroencephalography increases over occipital cortex. J
Neurosci. 2000; 20: Rc63.
- Haegens S, Luther L, Jensen O. Somatosensory anticipatory alpha
activity increases to suppress distracting input. J Cogn Neurosci.
2012; 24: 677-685.
- Cooper NR, Croft RJ, Dominey SJ, Burgess AP, Gruzelier JH. Paradox lost? Exploring the role of alpha oscillations during externally vs.
internally directed attention and the implications for idling and inhibition hypotheses. Int J Psychophysiol 2003; 47: 65-74.
- Cooper NR, Burgess AP, Croft RJ, Gruzelier JH Investigating evoked
and induced electroencephalogram activity in task-related alpha
power increases during an internally directed attention task. Neuroreport. 2006; 17: 205-208.
- Modi HN, Singh H, Yang GZ, Darzi A, Leff DR A decade of imaging surgeons’ brain function (part II): A systematic review of applications for technical and nontechnical skills assessment. Surgery
2017; 162: 1130-1139.
- Bernardi G, Ricciardi E, Sani L, Gaglianese A, Papasogli A, et al. How
skill expertise shapes the brain functional architecture: an fMRI
study of visuo-spatial and motor processing in professional racingcar and naïve drivers. PLoS One 2013:8:e77764.
- Bays PM, Gorgoraptis N, Wee N, Marshall L, Husain M Temporal
dynamics of encoding, storage, and reallocation of visual working
memory. Journal of vision. 2011; 11
- Bays BC, Visscher KM, Le Dantec CC, Seitz AR Alpha-band EEG activity in perceptual learning. J Vis. 2015; 15: 7.
- Li G, Li H, Pu J, Wan F, Hu Y Effect of brain alpha oscillation on the
performance in laparoscopic skills simulator training. Surg Endosc.
2021; 35: 584-592.
- Delorme A, Makeig S EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component
analysis. J Neurosci Methods. 2004; 134: 9-21.
- Lopez-Calderon J, Luck SJ ERPLAB: an open-source toolbox for the
analysis of event-related potentials. Front Hum Neurosci. 2014; 8:
213.
- Rouder JN, Morey RD, Speckman PL, Province JM Default Bayes
factors for ANOVA designs. 2012; 56: 356-374.
- Uemura M, Tomikawa M, Kumashiro R, Miao T, Souzaki R, et al.
Analysis of hand motion differentiates expert and novice surgeons.
J Surg Res. 2014; 188: 8-13.
- Guru KA, Shafiei SB, Khan A, Hussein AA, Sharif M, et al. Understanding Cognitive Performance During Robot-Assisted Surgery.
Urology. 2015; 86: 751-757
- Huffman E, Anton N, Martin J, Timsina L, Dearing W, et al. Optimizing Assessment of Surgical Knot Tying Skill. J Surg Educ. 2020; 77:
1577-1582.
- Graybiel AM Habits, rituals, and the evaluative brain. Annu Rev
Neurosci. 2008; 31: 359-387.
- Pfurtscheller G Event-related synchronization (ERS): an electrophysiological correlate of cortical areas at rest. Electroencephalogr
Clin Neurophysiol. 1992; 83: 62-69.
- Bauer M, Kluge C, Bach D, Bradbury D, Heinze HJ, et al. Cholinergic
enhancement of visual attention and neural oscillations in the human brain. Curr Biol. 2012; 22: 397-402.
- Foxe JJ, Simpson GV, Ahlfors SP Parieto-occipital approximately 10
Hz activity reflects anticipatory state of visual attention mechanisms. Neuroreport. 1998; 9: 3929-3933.
- Fu KM, Foxe JJ, Murray MM, Higgins BA, Javitt DC, et al. Attention-dependent suppression of distracter visual input can be cross-modally cued as indexed by anticipatory parieto-occipital alpha-
band oscillations. Brain Res Cogn Brain Res. 2001; 12: 145-152.
- Ben-Simon E, Podlipsky I, Okon-Singer H, Gruberger M, Cvetkovic
D, et al. The dark side of the alpha rhythm: fMRI evidence for induced alpha modulation during complete darkness. Eur J Neurosci
2013; 37: 795-803.
- Halgren M, Ulbert I, Bastuji H, Fabó D, Erőss L, et al. The generation
and propagation of the human alpha rhythm. Proc Natl Acad Sci U
S A. 2019; 116: 23772-23782.
- Floyer-Lea A, Matthews PM Distinguishable brain activation net-works for short- and long-term motor skill learning. J Neurophysiol. 2005; 94: 512-518.
- Leff DR, Leong JJ, Aggarwal R, Yang GZ, Darzi A Could variations in
technical skills acquisition in surgery be explained by differences in
cortical plasticity? Ann Surg .2008; 247: 540-543.
- Coynel D, Marrelec G, Perlbarg V, Pélégrini-Issac M, Van de Moortele PF, et al. Dynamics of motor-related functional integration
during motor sequence learning. Neuroimage. 2010; 49: 759-766.
- Krakauer JW, Mazzoni P Human sensorimotor learning: adaptation, skill, and beyond. Curr Opin Neurobiol. 2011; 21: 636-644.
- Balleine BW, Dezfouli A Hierarchical Action Control: Adaptive Collaboration Between Actions and Habits. Front Psychol. 2019; 10:
2735.
- Sauvage C, Jissendi P, Seignan S, Manto M, Habas C Brain areas
involved in the control of speed during a motor sequence of the
foot: real movement versus mental imagery. J Neuroradiol. 2013;
40: 267-280
- Shirinbayan SI, Dreyer AM, Rieger JW Cortical and subcortical areas involved in the regulation of reach movement speed in the
human brain: An fMRI study. Hum Brain Mapp. 2019; 40: 151-162.
- Pfurtscheller G, Neuper C, Krausz G Functional dissociation of lower and upper frequency mu rhythms in relation to voluntary limb
movement. Clin Neurophysiol. 2000; 111: 1873-1879.
- Button KS, Ioannidis JP, Mokrysz C, Nosek BA, Flint J, et al. Power
failure: why small sample size undermines the reliability of neuroscience. Nat Rev Neurosci. England. 2013; 365-376.