Introduction
RTs is a malignancy of urinary system, accounting for 2%-3%
of adult malignancies [1]. Although the prevalence of locally advanced RTs with inferior vena cava (IVC) tumor thrombus is relatively low, occurred in 4-10% of patients [2], tumor thrombi could
migrate proximally to the right atrium [3] and lead to poor prognosis. For RTs with IVC tumor thrombus, surgery could effectively
improve the prognosis [4]. As one of the most difficult operations
in urology, RAL-IVCTE has a complicated procedure, great operation trauma and a lot intraoperative bleeding. Therefore, it is necessary to keep patients in good condition as far as possible before
operation. However, in clinical observation, we found that RTs patients with IVC tumor thrombus had a high prevalence of preoperative anemia and great demand for allogeneic RBC transfusion.
Although several researches reported the high transfusion rate in
patients underwent RAL-IVCTE [5,6], studies on the effect of Hb
concentration on RTs patients with IVC thrombus who underwent
RAL-IVCTE are few, and lack of study on the effect of drug treatment of anemia for these patients, which is very important for
patient blood management.
As a kind of Erythropoiesis-Stimulating Agents (ESAs), rh-EPO
could improve the Hb levels in anemic patients. However, the use
of ESAs on surgical patients remains contrversial. Some studies
have reported the use of perioperative ESAs reduce the need for
allogeneic blood transfusion [7,8]. Other studies have found rh-EPO may lead increased thrombotic events [9,10]. Nevertheless,
the rh-EPO treatment influence for other clinically outcomes such
as ischemic events, thromboembolic events and mortality, the
number of events was too small and the variability in results was
too large to detect statistically significant and clinically relevant
differences [11]. In this study, we retrospectively analyzed the effects of rh-EPO administration on RTs patients underwent RAL-IVCTE.
Materials and methods
We retrospectively analyzed the data of 61 in-patients with RTs
who underwent RAL-IVCTE in the first medical central of Chinese
PLA General Hospital (PLAGH) from March 2014 to December
2018. Age less than 18 years old, unknown preoperative hemoglobin and laboratory data, and cases with unknown demographics were excluded. The study was approved by the Chinese PLAGH
ethics committee.
Demographic and clinical data including age, gender, body
mass index (BMI), smoking status in one year, drinking status
within 2 weeks, laboratory data, size of renal tumor, clinical stage,
IVC thrombus classification, Charlson comorbidity index (CCI), hypertension, American Society of Anaesthesiologists (ASA) score,
pathologic results were collected. Postoperative complications
were recorded according to the Clavien classification system[12].
Administration of rh-EPO during hospitalization was recorded,
10,000 international unit (IU) per dose. The decision and the
amount to use rh-EPO was by the preference of the treating surgeon. Transfusion was recorded in patients received at least one
unit of packed RBC transfused in perioperative period.
Anemia was defined as Hb <120 g/L for women and < 130 g/L
for men according to the 2011 WHO guidelines: no anemia (Hb
≥ 120 g/L in women and Hb ≥ 130 g/L in men), mild anemia (110
g/L ≤ Hb < 120 g/L in women and 110 g/L ≤ Hb < 130 g/L in men)
and moderate-to-severe anemia (Hb < 110 g/L). Patients’ last Hb
concentration before discharge was defined as discharge Hb concentration.
Statistical significance was defined as P <0.05, Confidence Intervals (CIs) are reported at the 95% level. Stata 15.1 was used to
analyze the data and draw graphics.
Patient demographics and baseline characteristics were analyzed using descriptive statistics and are presented as percentage
or mean ± SD. The student t test was used to examine the association between continuous variables, and Pearson χ2 test or the
Fisher’s exact test was used to examine the association between
categorical variables. We conducted several Multiple Linear Regression (MLR) analysis models. In these MLR analysis models, a
stepwise method was selected, treating intraoperative and post-operative RBC transfusion volume, days stay in ICU, postoperative
complication grade and the discharge Hb concentration, respectively, as the dependent variable and the following parameters
as independent variables including the last preoperative Hb concentration, rh-EPO injection dose preoperative, rh-EPO injection
dose postoperative, estimated blood loss, ASA grade, size of renal
tumor, IVC thrombus classification, TNM stage grouping, history
of targeted drug therapy (no, yes), affected kidney (right, left), pathology (other, clear cell ), tobacco use in past year (no, yes), alcohol intake within 2 weeks(no, yes), sex, age (continuous years),
BMI, CCI, white blood cell count, platelet count, international normalised ratio, blood urea nitrogen value, albumin value, aspartate
aminotransferase value, creatinine value.
In these MLR analysis models, marginal probabilities were calculated using Stata. Regression-predicted effects of preoperative
Hb concentration on intraoperative and postoperative RBC transfusion volume, and rh-EPO administration on days stay in ICU and
the discharge Hb concentration were plotted using marginal predictive plotting.
Results
We included data for 61 patients, 21 women (34.43%) and 40
(65.57%) men with a mean age of 55.8 years (SD 13.0, range 21.7
to 86.3). In the present study, a total of 41 patients (67.21%) had
anemia, of which 13 (21.31%) patients had mild anemia and 28
(45.90%) patients had moderate-to-severe anemia (including 1
patient had severe anemia). Table 1 shows the basic characteristics of patients. 43 (70.49%) patients received perioperative
RBC transfusion, including 42 (68.85%) patients received intraoperative and postoperative RBC transfusion and 1 (1.64%) patient
received preoperative RBC transfusion. 19 (31.15%) patients received perioperative rh-EPO treatment.
Table 1: Baseline characteristics of patients (n=61).
| Variables |
N (% or SD) |
| Age (yr) |
55.8 (13.0) |
| BMI (kg/m2) |
24.4 (3.2) |
| Gender |
|
| Female |
21 (34.43%) |
| Male |
40 (65.57%) |
| Preoperative Hb concentration (g/L) |
116.4 (20.7) |
| Preoperative anemia |
|
| Mild anemia |
13 (21.31%) |
| Moderate-to-severe anemia |
28 (45.90%) |
| Tobacco use in past year |
15 (24.59%) |
| Alcohol intake within 2 weeks |
9 (14.75%) |
| Hypertension |
18 (29.51%) |
| CCI≥1 |
22 (36.07%) |
| History of targeted drug therapy |
17 (27.87%) |
| Affected kidney |
|
| Right |
45 (73.77%) |
| Left |
45 (73.77%) |
| Pathology |
|
| Renal clear cell carcinoma |
41 (67.21%) |
| Other |
20 (32.79%) |
| Size of renal tumor (cm) |
7.9 (4.0) |
| TNM stage grouping |
|
| Stage III |
56 (91.80%) |
| Stage IV |
5 (8.20%) |
| IVC thrombus classification |
|
| Level I~ II |
29 (47.54%) |
| Level III~ IV |
32 (52.46%) |
| ASA grade |
|
| I- II |
32 (52.46%) |
| III~ IV |
29 (47.54%) |
| Estimated blood loss (1000ml) |
1.57 (2.17) |
| Total cost, K$ |
18.85 (7.75) |
| Intraoperative and postoperative RBC transfusion (%) |
42 (68.85%) |
| Perioperative rh-EPO treatment (%) |
19 (31.15%) |
In the MLR analysis of intraoperative and postoperative RBC
transfusion volume, lower the last preoperative Hb concentration
(r = -0.053; p = 0.025) and Charlson comorbidity index (r = -1.387;
p = 0.032), smaller size of tumor (r = -0.310; p = 0.010), and more
estimated blood loss (r = 3.859; p < 0.001) were related with more
intraoperative and postoperative RBC transfusion. Perioperative
rh-EPO therapy was not significantly associated with the need of
RBC transfusion (Supplementary Table S1). The analysis could account for the observed variation of the volume intraoperative and
postoperative RBC transfusion in 89.0% patients (R2 = 0.890).
Figure 1 shows the relationship of the last preoperative Hb
concentration and the intraoperative and postoperative RBC
transfusion volume. Figure 2 shows the relationship of the last
preoperative Hb concentration and the predicted intraoperative N; Number: SD; Standard Deviation: BMI; Body Mass Index: CCI; Charlson
Comorbidity Index: TMN; Tumor Node Metastasis: IVC; Inferior Vena
Cava: ASA; American Society Of Anaesthesiologists: Rh-EPO; Recombinant
Human Erythropoietin: RBC; Red Blood Cell. 1 unit RBC Was From 200ml
Whole Blood.
and postoperative RBC transfusion volume in MLR model. Lower
preoperative Hb level was significantly associated with more peri-operative RBC transfusion. The intraoperative and postoperative
transfusion RBC transfusion volume increased by 0.53 units for
every 10 g/L decrease in preoperative Hb concentration.
In the MLR analysis of the discharge Hb concentration, higher
preoperative Hb concentration (r = 0.383; p < 0.001), the first post-operative Hb concentration (r = 0.605; p < 0.001), preoperative
platelet count (r = 0.054; p = 0.003) and ASA grade (r = 9.194; p <
0.001); more intraoperative and postoperative RBC transfusion (r
= 0.602; p = 0.032) and preoperative rh-EPO injection (r = 3.306;
p = 0.014); lower postoperative complication grade (r = -3.966;
p = 0.039) were related with higher discharge Hb concentration
(Table 2). The analysis could account for the observed variation
of the discharge Hb concentration in 65.1% patients (R2 = 0.651).
Table 2: Multiple linear regression analysis of the discharge Hb concentration.
| The discharge Hb concentration |
r |
95% CI |
P |
Beta |
Adjusted R2 of the model |
| the last preoperative Hb concentration |
0.383 |
0.189 to 0.577 |
<0.001 |
0.434 |
0.651 |
| preoperative rh-EPO injection dose |
3.306 |
0.707 to 5.906 |
0.014 |
0.219 |
| Intraoperative and postoperative RBC transfusion volume |
0.602 |
0.054 to 1.150 |
0.032 |
0.295 |
| ASA grade |
9.194 |
4.258 to 14.129 |
<0.001 |
0.325 |
| IVC |
-3.407 |
-7.467 to 0.652 |
0.098 |
-0.165 |
| postoperative complication grade |
-3.966 |
-7.719 to -0.213 |
0.039 |
-0.251 |
| the first postoperative Hb concentration |
0.605 |
0.364 to 0.846 |
<0.001 |
0.637 |
| CCI |
-4.558 |
-9.370 to 0.253 |
0.063 |
-0.161 |
| the last preoperative platelet count |
0.054 |
0.020 to 0.088 |
0.003 |
0.278 |
CCI: Charlson comorbidity index: IVC: inferior vena cava: ASA: American Society of Anaesthesiologists; Hb: hemoglobin; rh-EPO: Recombinant human erythropoietin; RBC: red blood cell; CI: confidence interval.
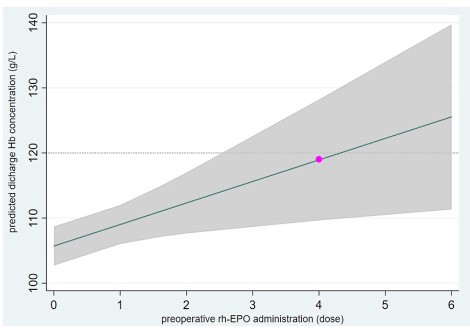
Figure 3 shows the relationship of the preoperative rh-EPO administration and the predicted discharge Hb concentration in MLR
model. When patients injected 4 doses rh-EPO preoperative, the
predicted median Hb could be 119 g/L (the red circle).
We graded complications of patients according to Clavien classification of surgical complications. In the MLR analysis of post-operative complication grade, higher Charlson comorbidity index
(r = 0.407; p = 0.011) and more intraoperative and postoperative RBC transfusion (r = 0.075; p < 0.001) were related with higher
postoperative complication grade (Table 3). Perioperative rh-EPO
injection was not significantly associated with higher complication
grade. The analysis could account for the observed variation of the
postoperative complication grade in 58.8% patients (R2 = 0.588).
There were 4 thrombotic events in patients, 1 in rh-EPO treatment patients, 3 in non-rh-EPO treatment patients. And there was
no significant difference between the two groups population (p =
0.085, data not shown).
Table 3: Multiple linear regression analysis of postoperative complication grade.
| Postoperative complication grade |
r |
95% CI |
P |
Beta |
Adjusted R2 of the model |
| Intraoperative and postoperative RBC transfusion volume |
0.075 |
0.049 to 0.101 |
<0.001 |
0.582 |
0.588 |
| IVC thrombus classification |
0.254 |
-0.008 to 0.515 |
0.057 |
0.194 |
| CCI |
0.407 |
0.098 to 0.716 |
0.011 |
0.228 |
| History of targeted drug therapy |
0.299 |
-0.033 to 0.631 |
0.076 |
0.160 |
| Tobacco use in past year |
-0.435 |
-0.881 to 0.012 |
0.056 |
-0.163 |
CCI: Charlson Comorbidity Index; IVC: Inferior Vena Cava; RBC: Red Blood Cell; CI: Confidence Interval.
In the MLR analysis of days stay in ICU, more preoperative rh-
EPO injection (r = -0.539; p = 0.012), smaller size of tumor (r =
-0.145; p = 0.022), lower grade of postoperative complication (r
= 1.055; p = < 0.001), IVC thrombus classification (r = 0.833; p =
0.008) and ASA (r = 1.235; p = 0.002) were related with less days
stay in ICU (Supplementary Table S1). The analysis could account
for the observed variation of the days stay in ICU in 54.6% patients
(R2 = 0.546).
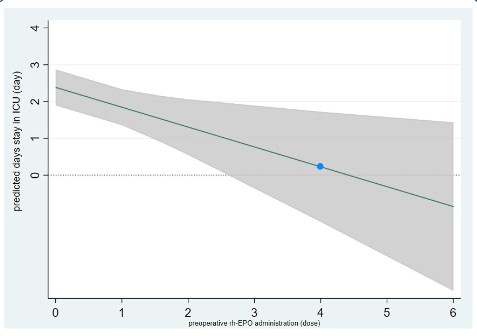
Figure 4 shows the relationship of the preoperative rh-EPO in-
jection dose and the predicted days stay in ICU in the MLR analy-
sis. The days stay in ICU decreased 0.5 day for every dose increase
of rh-EPO injection. When patients injected 4 doses rh-EPO pre-
operative, the predicted median days stay in ICU could be 0.2 day
(the bule circle).
Discussion
In this study, the incidence of preoperative anemia is very strik-
ing in RTs patients underwent RAL-IVCTE, 67.21% patients with
anemia, and 45.90% patients with moderate-to-severe anemia.
In present study, we found that patients lower preoperative Hb
concentration associated with more RBC transfusion. This result
was similar with a widely accepted finding that preoperative anemia increased the perioperative RBC transfusion demand. Rh-EPO
is one of the common agents to increase the Hb concentration in
clinic, and 31.15% patients received rh-EPO perioperative treatment in our study. We mainly analyzed the relationship of rh-EPO
administration and RBC transfusion, discharge Hb concentration, postoperative complication and days stay in ICU. As far as
we know, this is the first study of the impact of preoperative Hb
concentration and rh-EPO treatment on RTs patients underwent
RAL-IVCTE.
The 1-year disease-specific survival for untreated Renal Cell
Carcinoma (RCC) with venous tumor thrombus was 29% [13], but
the 1-year survival was 60% for surgery patients even with distant metastases at the time of diagnosis [14]. RTs patients with
IVC thrombus could benefit from a robot-assisted strategy and
achieve quick recovery [15]. RAL-IVCTE is highly intricated, even
surgeons with advanced robotic skill required a long operation
time [16]. One of the major risks of this surgery is bleeding, which
lead to a relatively high rate of blood transfusion. In our study, 42
(68.85%) patients received intraoperative and postoperative RBC
transfusion. The Hb concentration of patients was an important
factor relate to intraoperative and postoperative RBC transfusion
volume (Figure 1), with increased by 0.53 units for every 10 g/L
decrease in preoperative Hb concentration (Figure 2). However,
the preoperative and postoperative rh-EPO treatment were not
associated with RBC transfusion volume in this study. The negative
result may be due to the short rh-EPO treatment interval and relatively low dose of rh-EPO administration before operation. Among
the patients who used rh-EPO before operation, the time from
admission to surgery was 13.8 ± 3.5 days. However, Hb and hematocrit usually increased within 2-6 weeks after rh-EPO treatment.
Our result was similar with a prospective randomized multicenter
trial study in patients undergoing right hemicolectomy for carcinoma [17] which patients with preoperative rh-EPO treatment for
5-10 days, that rh-EPO not reduce the need of RBC transfusion.
In present study, our result showed that more preoperative
rh-EPO injection, rather than postoperative treatment, was significantly related with higher discharge Hb concentration. The increase of Hb is related to the improvement of patients' Quality of
Life (QOL) or fatigue. Several studies have found that changes in
Hb levels in patients with cancer and anemia caused by erythropoietin are associated with changes in their QOL [18-20], and the
greatest incremental QOL gain associated with a 10 g/L change
in Hb occurred around hemoglobin 120 g/L (range, 110-130 g/L)
[21]. Therefore, we recommend that clinicians should use rh-EPO as soon as possible for patients with preoperative anemia to
maintain a relatively high concentration of Hb in the perioperative
period.
Although a previous review showed that rh-EPO treatment in
RCC is not indicated for anemia in RCC is an infrequent clinical
problem [22], and also some studies assessing rh-EPO safety in
anemic patients with chronic renal disease have found adverse
effects including thromboembolic, cardiovascular and stroke
events, preoperative anemia in RTs patients with IVC is common,
and compared patients with chronic disease, the perioperative
dosing strategies of surgical patients are more variable in timing,
dose and duration [23,24]. In addtion, the effect of rh-EPO treatment on postoperative adverse events reminds controversial for
the quality of the evidence was not high due to inconsistency in
findings and small number of events were reported. Several studies found that rh-EPO therapy reduced the risk for acute kidney
injury [25], all-cause-mortality and end-stage renal disease[26],
and other studies have shown that rh-EPO administration did not
associated with an increased thromboembolic risk [27,28]. In this
study, we graded complications of patients according to Clavien
classification of surgical complications[12]. We found that preoperative treatment of rh-EPO not significantly associated with post-operative adverse complication grade. After that, we analyzed the
thrombotic events: 4 thrombotic events in patients, 3 in rh-EPO
treatment patients, 1 in non-rh-EPO treatment patients. And
there was no significant difference between the two groups population (p = 0.085, data not shown). Of note, in this study, we found
that more RBC transfusion was associated with higher grade post-operative complications. This is consistent with some large and
high-quality studies of the adverse effects of RBC transfusion on
surgical patients [29,30]. RTs patients underwent RAL-IVCTE have
a relatively high risk of bleeding and a high probability of allogeneic RBC transfusion. In our present study, rh-EPO treatment was
not significantly related to RBC transfusion requirements. However, from the perspective of the whole perioperative period, the
preoperative rh-EPO administration did improve the level of Hb in
patients. Clinicians should pay enough attention to preoperative
anemia in patients, give them treatment as soon as possible to
improve the Hb concentration. The early and adequate preoperative rh-EPO administration may indirectly reduce the demand for
perioperative RBC transfusion, and then reduce the occurrence of
postoperative complications.
We found in the present study that more preoperative, rather
than postoperative rh-EPO administration, was related with shorter ICU or postoperative hospital stay (data not shown), which was
similar to several other studies [31,32]. There may be several
reasons for this clinical benefit of rh-EPO. On the one hand, as a
hematopoietic hormone that regulates RBC production, rh-EPO
could improve the symptoms associated with anemia. On the other hand, in addition to stimulating hematopoiesis, rh-EPO can
also act as a tissue protectant with anti-inflammatory, cell stabilizing, and antiapoptotic effects on multiple organ systems including renal, cardiac and neurological systems [33,34]. However,
the minimum effective rh-EPO dose required to benefit patients
is unknown. In some previous reports, rh-EPO treatment protocols vary from multiple doses given over a period of 3 to 4 weeks
preoperatively at weekly intervals [35,36] to 10 days preoperative
daily given with extent to postoperative [37]. The United States
Food and Drug Administration approved a regimen of four subcutaneous injection of epoetin-α, 600 IU/kg of body weight, administered at 3, 2 and 1 week before surgery and again on the day of
operation [38]. In the present study, we found if patients injected
4 doses rh-EPO preoperative, the predicted median days stay in ICU could be 0.2 day (Figure 4), and the median discharge Hb con-
centration could be 119 g/L (Figure 3). Furthermore, we found it
was postoperative but not preoperative rh-EPO treatment associated with higher total hospital costs (Supplementary Table S1).
Considering this is a retrospective study, this finding could be due
to a selection bias such that patients received more postoperative rh-EPO treatment and less preoperative treatment, but this
is unlikely because there were 13 patients received preoperative
and 14 patients received postoperative rh-EPO treatment, and
the average injection doses were 2.4 and 2.9, respectively. Our
study indicated that early and adequate administration of rh-EPO
before operation can be beneficial to RAL-IVCTE patients. We recommend at least 4 doses preoperative rh-EPO administrate for
these patients.
Supplementary table S1: Multiple linear regression analysis of the factors associated with intraoperative and postoperative RBC transfusion
volume, days stay in ICU and total hospital fee.
|
r |
95% CI |
P |
Beta |
Adjusted R2 of the model |
| Intraoperative and postoperative RBC transfusion volume |
|
|
|
|
0.890 |
| the last preoperative Hb concentration |
-0.053 |
-0.099 to -0.007 |
0.025 |
-0.123 |
|
| BMI |
-0.262 |
-0.541 to 0.018 |
0.066 |
-0.093 |
|
| CCI |
-1.387 |
-2.648 to -0.127 |
0.032 |
-0.100 |
|
| Size of renal tumor |
-0.310 |
-0.542 to -0.077 |
0.010 |
-0.138 |
|
| estimated blood loss |
3.895 |
3.506 to 4.283 |
<0.001 |
0.946 |
|
| Days stay in ICU |
|
|
|
|
0.546 |
| IVC thrombus classification |
0.833 |
0.230 to 1.436 |
0.008 |
0.281 |
|
| ASA grade |
1.235 |
0.476 to 1.994 |
0.002 |
0. 304 |
|
| postoperative complication grade |
1.055 |
0.607 to 1.502 |
<0.001 |
0.465 |
|
| preoperative rh-EPO injection dose |
-0.539 |
-0.957 to -0.122 |
0.012 |
-0.250 |
|
| Size of renal tumor |
-0.145 |
-0.269 to -0.021 |
0.022 |
-0.221 |
|
| Total hospital fee |
|
|
|
|
0.811 |
| history of targeted drug therapy |
-2.077 |
-3.615 to -0.539 |
0.009 |
-0.165 |
|
| the last preoperative Hb concentration |
-0.040 |
-0.086 to 0.007 |
0.096 |
-0.106 |
|
| postoperative rh-EPO injection dose |
0.629 |
0.064 to 1.194 |
0.030 |
0. 142 |
|
| postoperative complication grade |
1.649 |
0.621 to 2.676 |
0.002 |
0.246 |
|
| days stay in ICU |
1.763 |
1.338 to 2.188 |
<0.001 |
0. 596 |
|
| IVC thrombus classification |
1.245 |
-0.0408 to 2.531 |
0.057 |
0. 142 |
|
| CCI |
-1.481 |
-2.977 to 0.015 |
0.052 |
-0.123 |
|
Conclusions
In conclusion, we found that preoperative Hb concentration in
RTs patients underwent RAL-IVCTE were relatively low and preop-
erative anemia was not uncommon among them. Patients with
lower Hb levels were associated with increased demand of intra-
operative and postoperative RBC transfusion. More preoperative,
rather than postoperative rh-EPO administration was related to
shorter ICU stay. The perioperative rh-EPO treatment was not
significantly associated with more RBC transfusion demand and
higher grade of postoperative complications.
Limitations
However, our study has several limitations. On the one hand,
we have a relatively small number of samples, which may be related to the relatively low incidence of RTs with IVC tumor thrombus.
On the other hand, we retrospective analysis the postoperative
complications during hospitalization, but not analysis the long-term complications and death rate of patients. Larger sample size
or prospective studies are needed to further verify our conclusion.
Declarations
Acknowledgment: This work was supported by the Bethune
Charitable Foundation (BQE-2018-SX-01) of China. Thanks to
the Bethune Charitable Foundation for funding this research.
We completed the study, including data collection, analysis and
interpretation. We were grateful for patient’s informed consent
and the data provided by the hospital. We declare that there are
no conflicts of interest. Our research was approved by the ethics committee of Chinese PLA General Hospital.
Funding: This work was supported by the Bethune Charitable
Foundation (BQE-2018-SX-01) of China.
Conflict of interest: The authors declare no conflict of interest.
Data availability statement: The data of this study are available by email of acada_ljting@sina.com.
References
- Wotkowicz C, Wszolek MF, Libertino JA. Resection of renal tumors
invading the vena cava. Urol Clin North Am. 2008; 35: 657-671.
- Marshall FF, Dietrick DD, Baumgartner WA, Reitz BA. Surgical management of renal cell carcinoma with intracaval neoplastic extension above the hepatic veins. J Urol. 1988; 139: 1166-1172.
- Hatcher PA, Anderson EE, Paulson DF, Carson CC, Robertson JE.
Surgical management and prognosis of renal cell carcinoma invading the vena cava. J Urol. 1991; 145: 20-23.
- Bansal RK, Tu HY, Drachenberg D, Shayegan B, Matsumoto E, et
al. Laparoscopic management of advanced renal cell carcinoma
with renal vein and inferior vena cava thrombus. Urology. 2014;
83: 812-816.
- Chopra S, Simone G, Metcalfe C, de Castro Abreu AL, Nabhani J,
et al. Robot-assisted Level II-III Inferior Vena Cava Tumor Thrombectomy: Step-by-Step Technique and 1-Year Outcomes. Eur Urol.
2017; 72: 267-274.
- Wang B, Huang Q, Liu K, Fan Y, Peng C, et al. Robot-assisted Level
III-IV Inferior Vena Cava Thrombectomy: Initial Series with Step-by-step Procedures and 1-yr Outcomes. Eur Urol. 2020; 78: 77-86.
- Bedair H, Yang J, Dwyer MK, McCarthy JC. Preoperative erythropoietin alpha reduces postoperative transfusions in THA and TKA
but may not be cost-effective. Clin Orthop Relat Res. 2015; 473:
590-596.
- Weltert L, Rondinelli B, Bello R, Falco M, Bellisario A, et al. A single
dose of erythropoietin reduces perioperative transfusions in cardiac surgery: results of a prospective single-blind randomized controlled trial. Transfusion. 2015; 55: 1644-1654
- Corwin HL, Gettinger A, Fabian TC, May A, Pearl RG, et al. Efficacy
and safety of epoetin alfa in critically ill patients. N Engl J Med.
2007. 357(10): 965-76.
- Stowell CP, Jones SC, Enny C, Langholff W, Leitz G. An open-label,
randomized, parallel-group study of perioperative epoetin alfa
versus standard of care for blood conservation in major elective
spinal surgery: safety analysis. Spine (Phila Pa 1976). 2009; 34:
2479-2485.
- Mueller MM, Van Remoortel H, Meybohm P, Aranko K, Aubron C,
et al. Patient Blood Management: Recommendations From the
2018 Frankfurt Consensus Conference. JAMA. 2019; 321: 983-997.
- Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004; 240: 205-213.
- Reese AC, Whitson JM, Meng MV. Natural history of untreated renal cell carcinoma with venous tumor thrombus. Urol Oncol. 2013;
31: 1305-1309.
- Whitson JM, Reese AC, Meng MV. Population based analysis of
survival in patients with renal cell carcinoma and venous tumorthrombus. Urol Oncol. 2013; 31: 259-263.
- Gu L, Ma X, Gao Y, Li H, Li X, et al. Robotic versus Open Level I-II
Inferior Vena Cava Thrombectomy: A Matched Group Comparative
Analysis. J Urol. 2017; 198: 1241-1246.
- Kundavaram C, Abreu AL, Chopra S, Simone G, Sotelo R,et al. Advances in Robotic Vena Cava Tumor Thrombectomy: Intracaval
Balloon Occlusion, Patch Grafting, and Vena Cavoscopy. Eur Urol.
2016; 70: 884-890.
- Kettelhack C, Hönes C, Messinger D, Schlag PM. Randomized multicentre trial of the influence of recombinant human erythropoietin
on intraoperative and postoperative transfusion need in anaemic
patients undergoing right hemicolectomy for carcinoma. Br J Surg.
1998; 85: 63-67.
- Duh MS, Lefebvre P, Fastenau J, Piech CT, Waltzman RJ. Assessing
the clinical benefits of erythropoietic agents using area under the
hemoglobin change curve. Oncologist. 2005; 10: 438-448.
- Lefebvre P, Vekeman F, Sarokhan B, Enny C, Provenzano R, Cremieux PY. Relationship between hemoglobin level and quality of
life in anemic patients with chronic kidney disease receiving epoetin alfa. Curr Med Res Opin. 2006; 22: 1929-1937.
- Stull DE, Vernon MK, Legg JC, Viswanathan HN, Fairclough D,
Revicki DA. Use of latent growth curve models for assessing the
effects of darbepoetin alfa on hemoglobin and fatigue. Contemp
Clin Trials. 2010; 31: 172-179.
- Shasha D, Cremieux P, Harrison L. Relationship between hemoglobin levels and quality of life during radiation therapy plus concomitant or sequential chemotherapy in patients with cancer and
anemia treated with epoetin alfa. J Natl Compr Canc Netw. 2004;
2: 509-517.
- Albers P, Heicappell R, Schwaibold H, Wolff J. Erythropoietin in urologic oncology. Eur Urol. 2001; 39: 1-8.
- Singh AK, Szczech L, Tang KL, Barnhart H, Sapp S, et al. Correction
of anemia with epoetin alfa in chronic kidney disease. N Engl J
Med. 2006; 355: 2085-2098.
- Pfeffer MA, Burdmann EA, Chen CY, Cooper ME, de Zeeuw D, et al.
A trial of darbepoetin alfa in type 2 diabetes and chronic kidney
disease. N Engl J Med. 2009; 361: 2019-2032.
- Penny-Dimri JC, Cochrane AD, Perry LA, Smith JA. Characterising the Role of Perioperative Erythropoietin for Preventing Acute
Kidney Injury after Cardiac Surgery: Systematic Review and Meta-Analysis. Heart Lung Circ. 2016; 25: 1067-1076.
- Oh SW, Chin HJ, Chae DW, Na KY. Erythropoietin improves longterm outcomes in patients with acute kidney injury after coronary
artery bypass grafting. J Korean Med Sci. 2012; 27: 506-511.
- Sowade O, Ziemer S, Sowade B, Franke W, Messinger D, et al. The
effect of preoperative recombinant human erythropoietin therapy
on platelets and hemostasis in patients undergoing cardiac surgery. J Lab Clin Med. 1997; 129: 376-383.
- Spahn DR, Schoenrath F, Spahn GH, Seifert B, Stein P, et al. Effect
of ultra-short-term treatment of patients with iron deficiency or
anaemia undergoing cardiac surgery: a prospective randomised
trial. Lancet. 2019; 393: 2201-2212.
- Abdelsattar ZM, Hendren S, Wong SL, Campbell DA Jr, Henke P.
Variation in Transfusion Practices and the Effect on Outcomes After Noncardiac Surgery. Ann Surg. 2015; 262: 1-6.
- Goel R, Patel EU, Cushing MM, Frank SM, Ness PM, et al. Association of Perioperative Red Blood Cell Transfusions With Venous
Thromboembolism in a North American Registry. JAMA Surg.
2018; 153: 826-833.
- Shapiro GS, Boachie-Adjei O, Dhawlikar SH, Maier LS. The use of
Epoetin alfa in complex spine deformity surgery. Spine (Phila Pa
1976). 2002; 27: 2067-2071.
- Shin HJ, Ko E, Jun I, Kim HJ, Lim CH. Effects of perioperative erythropoietin administration on acute kidney injury and red blood
cell transfusion in patients undergoing cardiac surgery: A systematic review and meta-analysis. Medicine (Baltimore). 2022; 101:
e28920.
- Brines ML, Ghezzi P, Keenan S, Agnello D, de Lanerolle NC, et al.
Erythropoietin crosses the blood-brain barrier to protect against
experimental brain injury. Proc Natl Acad Sci U S A. 2000; 97:
10526-10531.
- Coleman T, Brines M. Science review: recombinant human erythropoietin in critical illness: a role beyond anemia. Crit Care. 2004;
8: 337-341.
- Wurnig C, Schatz K, Noske H, et al. Subcutaneous low-dose epoetin beta for the avoidance of transfusion in patients scheduled
for elective surgery not eligible for autologous blood donation. Eur
Surg Res. 2001; 33: 303-310.
- Weber EW, Slappendel R, Hémon Y, Mähler S, Dalén T,et al. Effects
of epoetin alfa on blood transfusions and postoperative recovery
in orthopaedic surgery: the European Epoetin Alfa Surgery Trial
(EEST). Eur J Anaesthesiol. 2005; 22: 249-257.
- Kosmadakis N, Messaris E, Maris A, Katsaragakis S, Leandros E, et
al. Perioperative erythropoietin administration in patients with
gastrointestinal tract cancer: prospective randomized double-blind study. Ann Surg. 2003; 237: 417-421.
- Goldberg MA, McCutchen JW, Jove M, Di Cesare P, Friedman RJ, et
al. A safety and efficacy comparison study of two dosing regimens
of epoetin alfa in patients undergoing major orthopedic surgery.
Am J Orthop (Belle Mead NJ). 1996; 25: 544-552.