Introduction
In many human epithelial cancers there is an increased expression of Epithelial Growth Factor (EGF) and its receptor (EGFR).
Therefore, EGF/EGFR system is an interesting target for novel
anti-tumor therapy [1]. Passive immunotherapies with anti-EGFR
antibodies (mAb) are in clinical trials along or combined with conventional treatments, showing anti-tumor activities [2-4].
The epidermal growth factor ligand plays a very important role
in both, normal and pathological wound healing process; it promotes dermal wound healing stimulating proliferation and migration of keratinocytes. EGF also stimulates formation of granulation tissue and fibroblast motility [5].
Life expectancy is progressively increasing worldwide; advancing age is a very important factor for develops many types of cancers over the world. Thus, the demand of cancer cares, surgeries,
chemo-radiotherapies and immunotherapies are also increasing [6,7].
The relationship between ageing and wound healing had been
examined previously but there are contradictory criteria concerning the effect of age in wound healing; some authors conclude
that healthy aging people have a delayed cutaneous wound healing [8-10]. However others have evidenced a normal wound healing process in elderly people [11].
In the other hand, previous studies evidenced no impaired
wound healing process related to the EGF/EGFR blockade [12-14]. It is unknown whether anti-epidermal growth factor receptor
therapy possesses an additional risk for the wound healing process in aged patients. Thus, search to evaluate the effect of EGFR
block on the wound healing process of aged mice using the 7A7
mAb, a specific antibody against the murine EGFR [15].
Materials and methods
Ethics statement
All studies were conducted under a protocol approved by the
Institutional Animal Care and Use Committee from the National
Center for Laboratory Animal Breeding, with permit number
17/17.
7A7 mAb
7A7 mAb is an anti-murine EGFR extracellular domain monoclonal antibody (IgG1). It was generated by immunization of BALB/c
mice with the recombinant extracellular domain of murine EGFR
as a valuable tool for EGFR-based therapeutic pre-clinical studies in mice, in order to allow a more effective extrapolation of
the pre-clinical data to the clinical setting [15]. This mAb was also
described to prolong survival and show antimetastatic effects in a
D122 mouse tumor model [16].
Mice and immunization protocols
Female BALB/c/Cenp mice with 11-12 weeks and 53 or more
weeks of age were obtained from the National Center for Laboratory Animal Breeding (CENPALAB, Havana, Cuba) and maintained
in standard racks (Tecniplast, Varese, Italy). Autoclaved food EAO
1004 (CENPALAB, Havana, Cuba) and water were offered ad libitum. Room temperature (20–23oC), humidity (65 ± 10%) and the
photoperiod cycles (12 h per day), were automatically controlled.
Mice were treated with 2.8 mg/kg of 7A7 mAbor with Phosphate Buffered Saline (PBS) by intraperitoneal way on days 6,
4 and 2 before and days 2 and 5 after the skin wound. A group
of each age was maintained as wound healing control, without
treatment.
Wound healing model
All animals were anesthetized with intramuscular ketamine
chloride (50 mg/kg) (AICA, Havana, Cuba) and their dorsal regions
were depilated and washed with sodium chloride (NaCl) 0.9% (LA-BIOFAM, Havana, Cuba) and ethanol 70%. Then, 8 mm diameter,
full-thickness skin wound was performed on the back of each animal with a biotome (Acu Punch, Acuderm Inc., Fort Lauderdale.
In-life observations
The animals were monitored twice a day by an experienced
technician for any abnormal reactions, health problems or complications, and to determine if significant clinical abnormalities
were present in animals from any of the treatment groups. All
animals were weighed weekly using a precision balance (Sartorius, Germany).
Wound closure dynamics were measured with a caliper (Mitutoyo, Japan) at days 0, 2, 5, 8 and 12. Area was calculated by: π*a*b (a and b are radius of ellipse)
Digital photographs of the wounds were taken on days 8 and
13th after skin wound and the planimetry study was carried out
on skin image (2 images/animal). Digitalized images were treated
with the DIGIPAT IBM/PC computer system [17] and the following
parameters were determined:
1. Percent of total re-epithelized area, the percentage of
wound closure was calculated as: (area of original wound – area
of actual wound)/area of original wound × 100.
2. Percent of reduction in wound perimeter, was calculated as
(perimeter of original wound – perimeter of actual wound)/ perimeter of original wound × 100.
Histological preparation
A half of mice were euthanized at 8th day post-surgery and the
rest on the 13th day. Ulcer area and a portion of surrounding tissue were excised using surgical scissors. The samples were fixed
in 10% buffered formalin and paraffin-embedded sections were
stained with hematoxylin/eosin. Samples were blindly evaluated
by two pathologist for determining the extent of the healing process. Also, histological score for wound healing was determined
by two independent observers under an optical microscope using
semi-qualitatively graded as follow [18]:
Epidermis
Grade 1: Incomplete reepithelialization, scanty projection of
the epidermal edges with thin thickness.
Grade 2: Complete reepithelialization with thin epidermal
thickness and permanence of the desiccated clot.
Grade 3: Complete reepithelialization with moderate thickness
of the regenerated epidermis. Absence of the desiccated clot.
Dermis
Grade 1: Some collagen fibers in the neomatrix with no organization and focally distributed. The infiltration of macrophages and
angiogenesis is evident.
Grade 2: More presence of collagen fibers, partially orientated
in location parallelly to the epidermis. Persistence of some dilated
blood vessels.
Grade 3: Complete restitution of the new matrix with collagen fibers horizontally orientated. Absence of macrophages and
scanty collapsed blood vessels.
Statistical analysis
All statistical analyses were carried out using Minitab Data Analysis Program version 14 (Minitab Inc for Windows, 2003).
Statistical evaluation was performed by a randomized complete
Analysis of Variance (ANOVA) design with significance assessed
at p<0.05 level or by the unpaired t-test. When data did not have
a normal distribution, the Kruskall–Wallis test and the two-tailed
Mann Whitney test were used. The statistical evaluation of histological semi-qualitative analysis was performed by two-way
ANOVA.
Results
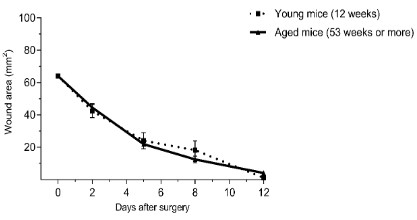
Ageing per se does not affect skin wound repair
Non-treated young and aging BALB/c mice with wounds in
their backs were observed daily and wound areas and planimetry
studies were performed at previously determined days. Figure 1
shows that wound closure dynamics between ages was similar.
Additionally, Table 1 shows there were no significant differences
between young and ageing mice according to the studied parameters.
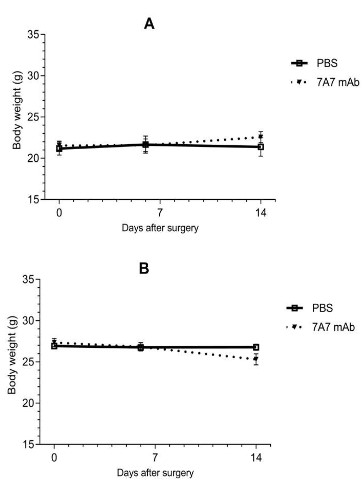
7A7 mAb was well tolerated in aged mice
No clinical signs were evidenced in mice during the whole experiment observational time period. No differences were seen in
body weight of immunized mice with full-thickness skin wound
respect to control animals, independently of age (Figure 2). Additionally, no changes appeared at the inoculation site in mice.
Skin wound repair in aged mice is not affected by EGFR block
A group of aged mice was immunized with 7A7 mAb or PBS as
control and a full-thickness skin wound was performed. As shown
in Figure 3, no significant differences were evidenced in wound
closure dynamics within age groups (p>0.05, Mann Whitney no
parametrical test).
During the hold study and after surgery no wound healing complications were observed in mice. In the histopathological study of
the resected skin displayed no complications of wound healing,
such as hyperplasia or changes in pigmentation in any animal.
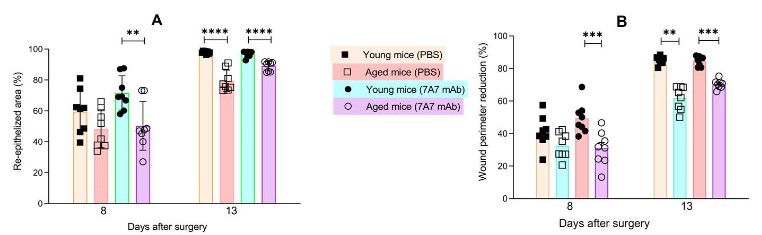
Wound healing is delayed in all aged treated mice compared
to young mice
Clinical observation of mice skin (Figure 4) suggests a delayed
wound healing in all aged mice, compared to young mice.
Planimetry study, using DIGIPAT software, on days 0, 8 and 13
after skin wound evidenced a significant reduction of studied parameters: percent of total re-epithelized area and percent of reduction in wound perimeter in all aged mice in comparison with
young mice, independently of the treatment (Figure 5).
Histopathological study on day 13 showed, in all young mice, a
corrected skin healing, independently of the received treatment;
with a completed wound re-epithelization, moderate thickness
epidermis, without scabs and a complete restitution of neo matrix in dermis, characterized by abundant horizontally oriented
without macrophages and sanguineous vessels. On the contrary,
in the 13th day after surgery, several aged animals showed scab
persistency in epidermis, collagen fibres with partial horizontal
orientation and abundant dilated sanguineous vessels in dermis.
Histological semi-qualitative analysis of wound repair (Table
2) and statistical analysis by two-way ANOVA corroborate no significant differences in wound healing between treatment inside
each age groups whereas the comparison between age according
therapy.
Table 1: Wound planimetry values 8 and 13 days after surgery in
young and aged BALB/c mice without treatment.
| Day |
Age |
Re-epithelized area (%) |
Wound perimeter reduction (%) |
Median |
Range |
Median |
Range |
| 8th |
Young |
49,00 |
37,10 |
28,60 |
31,50 |
| Aged |
51,90 |
62,32 |
42,50 |
23,50 |
| 13th |
Young |
98,075 |
0,585 |
86,98 |
0,76 |
| Aged |
93,54 |
3,58 |
75,19 |
5,15 |
Discussion
In response to tissue damage, the innate immune system responds trying to repair the tissue damaged integrity and its normal physiological functions [19]. During the aging process also
some intrinsic and extrinsic factors produce alterations, e.g. hormonal change levels, sun and contaminants exposition [20].
In the present study, no differences were evidenced in wound healing between young and aged mice without treatment. This result agrees with the current thinking that the effect of age af-
ter controlling known associated factors is not marked and that
wound healing in healthy older people is essentially normal
[11,21] despite alterations to individual processes.
Table 2: Histological semi-qualitative analysis of wound repair in
young and aged BALB/c mice with full-thickness skin wound (8 mm diameter) on the back, immunized with 7A7 mAb or maintained as
control (PBS).
| Day |
Age |
Treatment |
Epidermis (% of animals) |
Dermis (% of animals) |
1 |
2 |
3 |
1 |
2 |
3 |
| 8th |
Young mice |
PBS |
0 |
67 |
33 |
0 |
33 |
67 |
| AcM 7A7 |
67 |
0 |
33 |
33 |
33 |
33 |
| Aged mice |
PBS |
100 |
0 |
0 |
0 |
100 |
0 |
| AcM 7A7 |
100 |
0 |
0 |
0 |
100 |
0 |
| 13th |
Young mice |
PBS |
0 |
0 |
100 |
0 |
0 |
100 |
AcM 7A7 |
0 |
0 |
100 |
0 |
0 |
100 |
| Aged mice |
PBS |
0 |
60 |
40 |
0 |
60 |
40 |
| AcM 7A7 |
33 |
0 |
67 |
33 |
0 |
67 |
It is known that EGF ligand regulates many aspects of wound
healing, including inflammation, wound contraction, proliferation, migration, and angiogenesis. Previous studies about the
effects of anti-EGF/EGFR therapeutic anti-cancer drugs on the
wound healing process concluding that apparently, this kind of
treatment do not affect wound healing [12]. The present research
demonstrated no impact of EGFR block in skin wound repair, in
aged mice. A similar result was obtained by Fernández et al., who
in a retrospective study elucidated that old patients previously
depleted of EGF and receiving surgical procedures were without
post-surgical wound healing complications [14].
In spite of aged people has alterations in the normal skin process, it is not well documented that advanced age impairs wound
healing per se. Nevertheless, there is some inevitable injures in
old people that could produce local and systemic problems impairing wound healing [22].
There are many factors associated with the wound healing process such as pain and stress [23]. Contemporary evidences of different types of stress indicate an important impact on human and
animal wound healing [24,25]. Animals submitted to stress heal a
wound slow than control animals [26,27].
Neuro-hormonal mechanisms of stress originated from the
hypothalamus release hormones from the pituitary gland which
stimulates glucocorticoid hormone production, mainly cortisol in
the adrenal cortex [28]. Normal inflammatory and immune responses can be modified by stress due to a maintained release of
cortisol [29]. Therefore, a depression of some inflammatory cytokines (IL-1α, IL-1β, TNFα) and loss of matrix metalloproteinase
regulation interrupt the normal wound healing cascade [30,31].
Some authors had evidenced age-related changes in immune
cell populations and chemokine production during wound healing [32-34]. The results showed here evidenced the negative effect of repeated injections (PBS or 7A7 mAb) on aged mice wound
healing. It corroborates the harmful effect of stress and age which
should be related with reduction in the IL-1 producing capacity by
cells of old mice associated with immunosenescence [35]; this factor, joined to age-related alterations of collagen fibrils, impaired
skin structure and function [36] and delayed angiogenesis [32,37]
creates a tissue microenvironment that promotes delayed wound
healing in stressed aged individual. However, specific mechanisms
related with delayed wound healing in aged stressed individuals
were not evaluated in the present study, which is a limitation for
this study. Further research need to be conducted to investigate
the relationship between physiological mechanisms underlying
delayed wound healing in aged individuals and its relation with
stress.
Conclusion
In summary, our data showed that aging or EGF block per se
had not a deleterious effect on the healing process in adult mice
but stress and aging combination can affect significantly wound
healing.
Declarations
Data availability: The data that support the findings of this
study are available from the corresponding author on request.
Author contributions: All authors contributed to the study
conception and design:
Conceptualization: Dasha Fuentes, Angel Casacó
Data Curation: Daniel Jay, Nidia Fernández
Investigation: Dasha Fuentes, Daniel Jay, Nidia Fernández, Belinda Sánchez
Methodology: Dasha Fuentes, Angel Casacó, Nidia Fernández
Supervision: Angel Casacó
Writing – Original Draft Preparation: Dasha Fuentes
Writing – Review & Editing: Angel Casacó, Belinda Sánchez
All authors read and approved the final manuscript.
Conflict of interest: The authors declare that they have no conflicts of interest.
Acknowledgments: Not applicable.
Funding statement: Not applicable.
References
- Salomon DS, Brandt R, Cardiello F, Normanno N. Epidermal growth
factor–related peptides and their receptors in human malignancies. Crit Rev Oncol Hematol. 1995; 19: 183-232.
- Boerner JL, Danielsen A, Maihle NJ. Ligand-independent oncogenic signaling by the epidermal growth factor receptor: v-ErbB as a
paradigm. Exp Cell Res. 2003; 284: 111-121.
- Crombet T, Osorio M, Cruz T, Roca C, del Castillo R, et al. Use of
the humanized anti-epidermal growth factor receptor monoclonal
antibody hR3 in combination with radiotherapy in the treatment
of locally advanced head and neck cancer patients. J Clin Oncol.
2004; 22: 1646-1654.
- Xiong HQ, Rosenberg A, LoBuglio A, Schmidt W, Wolff RA, Deutsch
J, et al. CetuximAb, a monoclonal antibody targeting the epidermal
growth factor receptor, in combination with gemcitabine for advanced pancreatic cancer: a multicenter phase II Trial. J Clin Oncol.
2004; 22: 2610-2616.
- Carpenter G, Cohen S. Epidermal growth factor. J Biol Chem. 1990; 265: 7709-7712.
- Yancik R. Population aging and cancer: a cross-national concern.
Cancer J. 2005; 11: 437-441.
- Smetana K, Lacina L, Szabo P, Dvořánková B, Brož P, et al. Ageing as
an important risk factor for cancer. Anticancer Research. 2016; 36: 5009-5018.
- Guo S. and DiPietro LA. Factors Affecting Wound Healing. J Dent
Res. 2010; 89: 219-229.
- Soybir OC, Gurdal SO, Oran ES, Tulubas F, Yuksel M, et al. Delayed
cutaneous wound ̇healing in aged rats compared to younger ones.
Int Wound J. 2012; 9: 478-487.
- Sgonca R, Gruber J. Age-related aspects of cutaneous wound healing: a mini-review. Gerontology. 2013; 59: 159-164.
- Minimas DA. Ageing and its influence on wound healing. Wounds
UK. 2007; 3: 42-50.
- Casacó A, Fuente D, Ledón N, Fernández A, Crombet T. Anti-epidermal growth factor/epidermal growth factor receptor therapeutic anti-cancer drugs and the wound healing process. J Cancer Sci
Ther. 2012; 4: 324-329
- Fuentes D, Chacon L, Casaco A, Ledon N, Fernandez N, et al. Effects
of an EGFR-based cancer vaccine in the wound healing and inflammation processes in murine experimental models. Int Wound J.
2012.
- Fernández-Lorente A, Acosta- Brooks S, Neninger-Vinageras E, Barroso MC, Wilkinson B, Troche M, et al.Effect of blockade of the EGF
system on wound healing in patients vaccinated with CIMAvax®
EGF. World J Surg Onc. 2013; 11: 275.
- Garrido G, Sánchez B, Rodriguez HM, Lorenzano P, Alonso D, Fernandez LE et al. 7A7 mAb: A New Tool for the PreClinical Evaluation
EGFR-Based Therapies. Hybrid Hybridomics. 2004; 23: 168-175.
- Garrido G, Rabasa A, Garrido C, López A, Chao L, García-Lora AM,
et al. Preclinical modeling of EGFR-specific antibody resistance:
oncogenic and immune-associated escape mechanisms. Oncogene. 2014; 33: 3129-3139.
- Coro RM, Borrajero I. DIGIPAT. Un sistema cubano para morfometría de imagenes. RevLatinoam Patol. 1996; 34: 9-10
- Berlanga J, Moreira E, Perez L, Boix E, Gonzalez T, et al. Wound
healing promotion in rats treated with EGF is dose dependent. Biotecnologia Aplicada. 1996; 13: 181.
- Shaw TJ, Martin P. Wound repair at a glance. J Cell Sci. 2009; 122: 3209-3213.
- Zouboulis CC, Makrantonaki E. Clinical aspects and molecular diagnostics of skin aging. Clin Dermatol. 2011; 29: 3-14.
- Ashcroft GS, Horan MA, Ferguson MWJ. Ageing and Cutaneous
WoundHealing. In: MA Horan and RA Little (eds) Injury in the Ageing. Cambridge UniversityPress, Cambridge. 1998: 147-153
- Van De Kerkhof PCM, Van Bergen B, SpruijtK, Kuiper JP. Age related
changes in wound healing. Clin Exp Dermatol. 1994; 19: 369-374.
- Woo KY. Meeting the challenges of wound-associated pain: anticipatory pain, anxiety, stress, and wound healing. Ostomy Wound
Manage. 2008; 54: 10-12.
- Charalambous C, Vassilopoulos A, Koulouri A, Eleni S, Popi S, et al.
The Impact of stress on pressure ulcer wound healing process and
on the psychophysiological environment of the individual suffering
from them. Med Arch. 2018; 72: 362-366.
- Winn M, Holloway S. The impact of psychological stress on wound
healing: a theoretical and clinical perspective. Wounds UK. 2019;
15: 20-27.
- Padgett DA, Marucha PT, Sheridan JF. Restraint stress slows cutaneous wound healing in mice. Brain Behav Immun. 1998; 12: 64-73.
- Hübner G, Brauchle M, Smola H, Madlener M, Fässler R, et al. Differential regulation of pro-inflammatory cytokines during wound
healing in normal and glucocorticoid-treated mice. Cytokine.
1996; 8: 548-556.
- Bomholt S, Harbuz M, Blackburn-Munro G, Blackburn-Munro R.
Involvement and role of the hypothalamo-pituitary-adrenal stress
axis in animal models of chronic pain and inflammation. Stress.
2004; 7: 1-14.
- Webster Marketon J, Glaser R. Stress hormones and immune function. Cell Imm. 2008; 252: 16-26.
- Abraham DJ, Shiwen X, Black CM, Sa S, Xu Y, et al. Tumor necrosis
factor alpha suppresses the induction of connective tissue growth
factor by transforming growth factor-beta in normal and scleroderma fibroblasts. J Biol Chem. 2000; 275: 15220-15225.
- Caley MP, Martins VL, O’Toole EA. Metalloproteinases and Wound
Healing. Adv Wound Care (New Rochelle). 2015; 4: 225-234.
- Swift ME, Burns AL, Gray KL, DiPietro LA. Age-related alterations
in the inflammatory response to dermal injury. J Invest Dermatol.
2001; 117: 1027-1035.
- Ashcroft GS, Mills SJ, Ashworth JJ. Ageing and wound healing. Biogerontology. 2002; 3: 337-345.
- Gupta S. Molecular mechanisms of apoptosis in the cells of the immune system in human aging. Immunol Rev. 2005; 205: 114-129.
- Inamizu T, Chang MP, Makinodan T. Influence of age on the production and regulation of interleukin-1 in mice. Immunology.
1985; 55: 447-455
- Quan T, Fisher G, J. Role of age-associated alterations of the dermal extracellular matrix microenvironment in human skin aging: a
mini-review. Gerontology. 2015; 61: 427-434
- Ashcroft GS, Horan MA, Ferguson MW. Aging is associated with
reduced deposition of specific extracellular matrix components,
an upregulation of angiogenesis, and an altered inflammatory response in a murine incisional wound healing model. J Invest Dermatol. 1997; 108: 430-437.