Introduction
Healing shoulder joints remains a clinical, scientific, and societal challenge. Total Shoulder Arthroplasty (TSA) or hemi-arthroplasty (HA) represent the main current healing surgeries to
replace a wounded shoulder. TSA presents good short-terms clinical outcomes clinical, but shows an increasing risk for the glenoid
component failure after mid-terms follow-up on young patients
[1,2]. HA is a more conservative surgery procedure that saves
the patient native glenoid cavity. Still, a 17-years follow up clinical study showed that 75% of the patients who underwent a HA
did suffer from pain due to glenoid erosion [3]. Glenoid erosion is
characterised by a loss of glenoid bone stock. Clinically, it is generally explained by the degeneration of the cartilage and the wear of
the underlying bone due to detrimental tissue-implant frictions.
Current hemi-implants are mainly made of cobalt chrome (CoCr),
that is known to have bad wear outcomes against cartilage compared to other biomaterials [4-6]. Recently, hemi-implants with a
pyrocarbon (or pyrolytic carbon, PyC) head have shown interesting short-terms and mid-terms outcomes [7,8]. PyC is known to
have enhanced wear characteristics compared to CoCr [5,9], what
can partly explain a lower glenoid erosion rate [10].
The joint biomechanics is of great importance in its integrity.
Bone and cartilage are both biologically sensitive to the applied
mechanical loading. More particularly, it is assumed that the
mechanical fatigue-induced bone remodeling is involved in cartilage degeneration [11,12]. It is also known that the subchondral
bone marrow stimulation influences the cartilage integrity. Marrow stem cells from have a chondrogenic capacity [13] and bone
marrow lesions due to an abnormal loading have been associated with cartilage degeneration [14]. Monitoring the mechanical
loading transmitted to the bone is thus determinant in the regulation of the cartilage layer. In regard of this bone-cartilage cross
talk, a method consisting in drilling microholes in the subchondral
plate to promote bone-cartilage communication was developed
for the treatment of osteoarthritic joint [15]. This microdrilling
is assumed to liberate stem cells and other elements from the
underlying bone marrow thus promoting new cartilage formation
[16]. Interestingly, phospholipids (PL) are assumed to play a major role in cartilage surface lubrication. by being adsorbed on the
cartilage PL increases the surface hydrophobicity thus limiting its
interactions with other tissues [17]. Furthermore, the amount of
PL on osteoarthritic cartilage surface is decreased together with
its hydrophobicity [17]. The increased interactions between the
cartilage with an increased hydrophilicity with surrounding tissues partly explain its degeneration. Promoting the formation of
a lubricant layer at the glenoid implant interface may thus be a
relevant way to decrease tissue-implant mechanical frictions and
the development of durable treatments. Interestingly, bone marrow is known to contain phospholipids [18]. In that context, the
current study aim was to investigate the differences in the nature
of human glenoid tissues in contact with hemi-implant heads in
different HA configurations. These tissues were harvested from
three HA clinical cases: a CrCo hemi-implant head (CrCo), a PyC
hemi-implant head (PyC), and a PyC hemi-implant head with
subchondral microdrilling (μPyC). The tissues were investigated
through histological and lipidomic analyses.
Materials and methods
Cohort
The cohort consisted in 3 patients who underwent a revision
surgery after a HA between 2016 and 2021 by three different surgeons. The tissue samples were recovered with the patients and
the surgeons’ consent.
• Case 1: CrCo head (CrCo)
This patient was a 60 y.o. man at the time of the HA revision.
He had a previous surgery consisting of staples implantation to
stabilize the joint (unknown date of surgery). On March 2015, he
underwent a CrCo HA surgery because of abnormal pain and osteoarthritis of the joint. Because of the glenoid erosion against
the metallic head, the patient suffered from pain and underwent
a revision surgery after 21 months (December 2016). The surgeons diagnosed that due to the misplacement of the implant, osteophytes appeared without glenoid deviation. The HA was then
converted in a total reversed shoulder arthroplasty.
• Case 2: PyC head (PyC)
This patient was a 59 y.o. man at the time of the HA revision.
The PyC head was implanted in November 2018 and revised 31
months after (June 2021) due to pain. The surgeons did not identify accurately the link between the device and the pain. After head
removal, the surgeon noticed a glenoid cartilage in good status.
The hypothesis was that the implant stem was oversized. The HA
was converted in to total shoulder arthroplasty.
• Case 3: PyC hemi-implant with microdrilling (μPyC)
This patient was a 67 y.o woman at the time of the HA revision.
In 1991, she suffered from a traumatic humeral head fracture and
was treated using plate osteosynthesis to treat tuberosity fractures. In September 2015, she underwent a HA using a PyC head
because of pain and high glenoid erosion due to traumatic osteoarthritis. Microdrilling were performed by the surgeon on the areas lacking cartilage. the μPyC HA was revised 15 months later
(January 2017) due to abnormal pain. The surgeon declared that
the patient presented a malunion after the humeral head trauma making it difficult to place the hemi-implant stem properly.
In consequence, the stem was lateralized, generating abnormal
hypertension in the joint.
Tissue removal
The investigated tissues were harvested directly by the surgeons at the time of the HA revision. It was told to the surgeons to
harvest both a part of the glenoid cartilage membrane and a bone
core (Figure 1). For the PyC case (case 2), the surgeons harvested
a bone-cartilage core. The tissues were then stored in AFA (Alcool-Formol-Acetic Acid) until preparation for histological analyses.
Tissue analysis
Samples were fixed in 4 v% formalin, embedded in paraffin,
and 5 μm sections were cut. Mineralized tissues were decalcified
before cutting. Hematoxylin, Eosin, Safran (HES) staining was then
applied on different sections. HES stains cells nuclei in purples,
cells cytoplasm in pin and extracellular matrix in yellow.
Additionally, immunochemical stainings were applied to analyse the expression of collagen I (Coll I) and collagen II (Coll II) in
the samples. After removing the paraffin, the samples were immerged in 0.5% hyaluronidase buffered in PBS-BSA 3% for 1h at
room temperature in order to expose the antigenic sites. Sections were then incubated with rabbit anti-human Coll I diluted
at 1/1000 (20111, Novotec, France) and rabbit anti-human Coll
II diluted at 1/500 (20211, Novotec, France) overnight at 4°C
buffered in PBS-BSA 3%. Samples were then incubated with goat
anti-rabbit secondary anti-body coupled with peroxidase (K4002,
Envison Lapin, Dako, USA). A final reaction with diaminobenzidine
(K36008, Dako, USA) revealed the antigenic – antibody complexes
through a brown color. All the samples were analysed by Novotec
(Lyon, France) except from the HES of the PyC case that was analyse by the Ciqle platform (Lyon, France).
Lipidomic analysis
The CrCo and PyC explanted heads were first washed with saline solution to remove traces of blood. A solution of ethanol and
chloroform (1:2, v:v) was then used to wash twice the heads and
extract the lipids. The solutions were then stored at – 20°C until
analysis. Before the analysis, the samples were dried and diluted
in 100 μL of the same solvent (ethanol: chloroform, 1:2). Lipids
were separated through thin-layer chromatography. In brief, 90
μL of the solution were deposited on a thin-layer chromatography plate silica gel (60 F254, Merck). The migration occurred in a
mixture of n-hexane, diethyl ether, and acetic acid (80:25:1, v:v:v)
for 90 minutes. Samples were then dried under dry nitrogen;
migration spots were visualized under UV lamp at 254 nm and
compared with standards. The identified phospholipids (PL) were
then collected and diluted in a solution of 1 mL of methanol and
toluene (1:1, v:v) and 500 μl of 14% boron-trifluoride in methanol.
The solutions were then heated at 100°C for 90 minutes to allow
for transesterification that separates the fatty acids from the lipid
heads. The fatty acids were then extracted using 2 mL isooctane.
The remaining lipids solutions were centrifuged 5 minutes at 2000
rpm at 25°C to obtain a tri-phasic separation. The supernatant organic phase was removed. This step was performed three times.
Dried samples were diluted in 100 μL isooctane for gas chromatography. The PL concentration (nmol/mL) was then normalized
over the implant surface (nmol/mL/mm²).
Results
Tissue analysis
The general appearance of the sample harvested from the different cases is shown in the supplementary materials for each
staining and immunostaining (figures S1 to S5). Details are provided below.
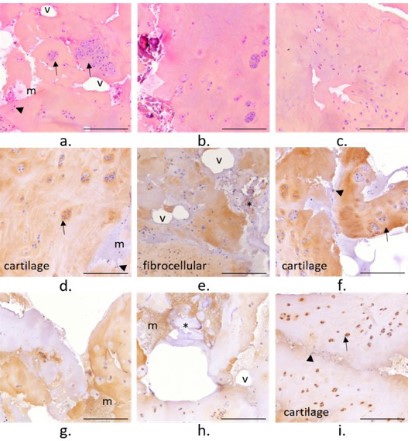
• CrCo
The membrane harvested from the clinical case 1 with an implanted CrCo head appeared as a loose and multi-layered tissue
(Figure S1). The tissue presented areas with hypertrophic chondrocytes (➔). Large clusters of these chondrocytes were also
observed. Large areas of low cell density were observed. Some
mineralized areas were observed (*, Figure 2b, e, and h) together
with a bone marrow or haemorrhagic type matrix (m). Sparse
black particles were also observed over the membrane (►, Figure 2a, f, and i). Coll I was more expressed over the membrane
compared to Coll II. The areas with chondrocytes or chondrocytes
clusters were mainly composed of Coll I, suggesting that the tissue is mainly a fibrocartilaginous tissue. Still, some Coll II was expressed intracellularly. The tissue was highly vascularized all over
the sample (v) (Figure 2).
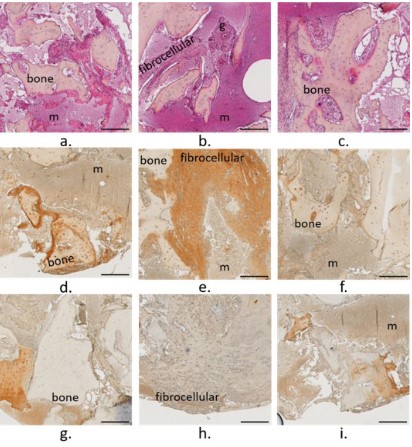
The bone core harvested from the CrCo case presented a porous bone network (Figure S2). Bone marrow was characterized
as a dense and hypercellular marrow (m) [19] that presented a
fibrocellular in some area with a high expression of Col l, highlighting an inflammation of the marrow (Figure 4). Granulation tissue
was also observed in the bone marrow (Figure 4b).
• PyC
The membrane harvested from the clinical case 2 with a PyC
head but no microdrilling appeared as a thick and dense membrane (Figure S3). The tissue was mainly made of a fibrocellular
and vascularized tissue (v) mainly composed of Coll I. Still an
amorphous region filled with chondrocytes (➔, Figure 3a) and
composed of Coll II (Figure 3g, and h) highlighted the presence
of an articular cartilage-like tissue. It has to be noticed that Coll II
was expressed on the vascular canals surface (Figure 3). The bone
marrow (m) appeared less dense than for the previous case. But
unfortunately, the core harvested by the surgeon did not presented a large quantity of bone tissue.
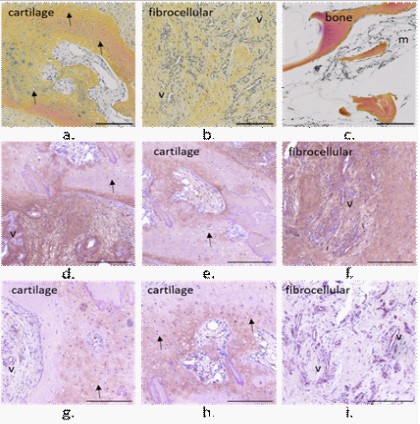
• μPyC
The membrane harvested from the clinical case 3 with a PyC
head and subchondral microdrills appeared as a dense a thick
membrane (Figure S4). The tissue presented an area of fibrocellular tissue surrounded by cartilaginous tissue. The tissue on the
bone side appeared vascularized (v) whereas on the implant side
a cartilaginous tissue with chondrocytes (➔) was observed (Figure 5). Still on the implant side, a dense layer highlight expressed
in Coll II was observed ({). Coll I and II were expressed intensively
in different areas, and also within the chondrocytes pericellular
matrices (Figure 5e, h, and i). Clusters of two chondrocytes oriented perpendicularly to the membrane surface were observed
(Figure 5e and h). Coll II was expressed on the vascular canal surfaces (Figure 5i).
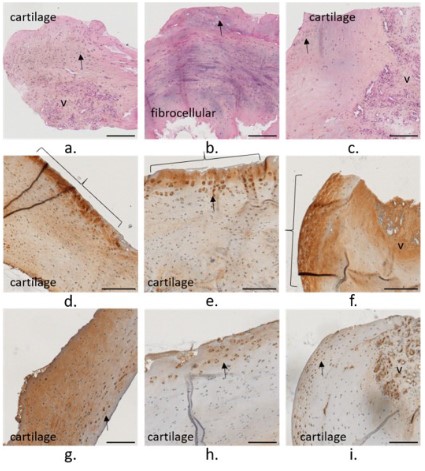
The bone core presented a more compact region on the implant side and a porous one away from the implant (Figure S5). A
low-density bone marrow (m) was observed with some sparse region of fibrocellular matrix (Figure 6). Coll I was mainly expressed
in the bone marrow, and Coll II was expressed on the surface of
vascular canals (v) (Figure 6h and i).
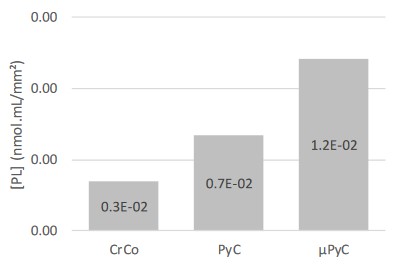
Lipidomic analysis
The concentration of phospholipids (PL) extracted from the
hemi-implant heads was lower on the CrCo head compared to the
PyC, with a higher concentration obtained on the μPyC head after
microdrilling in the subchondral bone (Figure 7).
Discussion
Shoulder hemi-arthroplasty (HA) currently represents an interesting way to maintain the mobility for the treatment of osteoarthritis or humeral trauma. Still, the complex frictional interaction
between the hemi-implant head and the native glenoid cavity may
result in glenoid erosion, decreasing the patient quality of life and
increasing the risk for the implant revision. In the current study,
the glenoid tissues (cartilage and bone) harvested from three clinical cases that underwent different natures of HA (CrCo head, PyC
head, and PyC head with subchondral microdrills) were characterized through histology. The results showed that the nature of the
glenoid tissues depended on the nature of the HA protocol. The
case with the CrCo head presented the worst tissue. On the other
hand, the case with the PyC head and subchondral microdrills appeared as the best case. While the membrane harvested from the
CrCo case presented a large area of cartilage, this cartilage was
of bad quality, as highlighted by the presence of large clusters of
chondrocytes [20] and of acellular areas [21]. Furthermore, the
membrane was mainly composed of Coll I, that is not a biomarker
for an healthy articular cartilage, but more of a fibrocartilaginous
tissue [22]. The presence of haemorrhagic areas supported the
inflammation of this glenoid cartilage membrane.
In contrast, a region of articular-like cartilage was observed on
the membrane harvested from the PyC case. Even if this region
was surrounded by a vascularized fibrocellular tissue, the formed
cartilage in this membrane was of good quality according to this
histological analysis. It is also interesting to observed Coll II on the
vascular canals surface. Coll II, synthetized by the chondrocytes, is
a biomarker for articular cartilage. Observing Coll II in the vascular
network suggests that the suitable biological signal for articular
cartilage synthesis has been sent to the vascularized subchondral
bone.
In the μPyC case, the tissue looked like an articular cartilaginous-like tissue, except for a vascularized area and a fibrocellular
region on the bone side. The dense layer on the implant sided
reminded of the dense layer on the cartilage surface [23] supporting a good sliding interactions between the PyC implant on the
glenoid cartilage membrane [24].
It is interesting to see that the nature of these different glenoid cartilage membranes was in accordance with the features
observed in the bone cores, more particularly in the bone marrow. The hypercellular and fibrous tissue observed for the CrCo
case support an inflammatory state of the bone marrow. Furthermore, the granulation tissue is also in accordance with a degenerative cartilage [25]. Conversely, the morphology of the bone
marrow observed in the μPyC case is more representative of a
healthy state of the subchondral bone [25]. It is also interesting
to observed the expression of Coll II in the bone marrow for the
μPyC, suggesting that a chondrogenesis signalling was transmitted to the chondrogenic bone marrow cells [26]. Unfortunately,
only a small part of bone is seen on the bone-cartilage core harvested from the PyC case. Still, the small region of bone marrow
presented a morphology closer to that from the μPyC case. Bone
marrow lesions are known to be associated with cartilage degeneration, reflecting the damaged state of the subchondral bone
[14]. Hence, these results strongly suggest the influence of the
hemi-implant material and the HA procedure on the biomechanical loading transmitted to the subchondral bone. This hypothesis
is further supported by the lipidomic analyses showing a higher
amount of PL adsorbed to the PyC surface compare to the CrCo.
This results is in accordance with previous ex vivo analyses showing a better adsorption of PL on PyC during friction on cartilage
[9]. Similar to the native cartilage, this higher concentration of PL
may decrease the friction interactions between the PyC and the
native glenoid tissues. This in turns decrease the frictional forces
and promote the sliding between the PyC implant and the native
cavity. In addition to an improve mechanobiological behaviour of
the implanted joint, a decrease frictional force may also prevent
from implant particles to be released in the contacting tissue, as it
was observed in the tissue harvested from the CrCo case. The PyC
better capacity to adsorbed a layer of PL may explain the better
clinical outcomes obtained with PyC HA compared to CrCo HA.
Interestingly, the concentration of PL on the μPyC with subchondral microdrills was higher than the PyC alone. This supports
the liberation of PL from the bone marrow and their diffusion toward the implant surface. Hence, in addition to the releasing of
chondrogenic stem cells from the bone marrow, microdrills may
also improve the biomechanical interactions within the implanted
joint.
This study is limited by a low number of cases, with 1 case per
HA configuration. It has to be noticed that PyC has only recently
been used for shoulder HA [10] and the number of PyC HA revision
remains low. The current study has been initiated in the 2015’s by
our groups after the observation of a neosynthtized cartilaginous
like tissue at the bone-pyrocarbon interface by orthopaedic surgeons. A large collection of these tissue explanted at the time of
a PyC HA revision is undergoing. The current study supports the
relevance of such collection to better understand the influence
of PyC on the integrity of the glenoid cartilage. More particularly,
the current study strongly suggest that the PyC is able to send
a suitable signal to the subchondral bone to promote cartilage
integrity regulation. Amongst others, this signalling seems to be
sent with the suitable biomechanical configuration offered by the
couple PyC – phospholipids, promoting the lubrication of the PyC
glenoid cavity lubrication (Figure 8).
Conclusion
In the current study, the glenoid cartilage and bone harvested from 3 clinical cases who underwent three different shoulder
hemi-arthroplasties: a case with a cobalt-chrome head, a second
with a pyrocarbon head, and a last with a pyrocarbon head and
subchondral microdrills. The histological and lipidomic analyses
showed better quality cartilage and bone tissues of better quality
for the pyrocarbon head with subchondral microdrills. Pyrocarbon
capacity to adsorb a phospholipids layer on its surface associated
with the liberation of bone marrow phospholipids through the microdrills may improve the biomechanical interactions at the cartilage implant interface. Despite the limitations of this study, the
results obtained strongly support the interest to further analyse
the pyrocarbon glenoid cavity biomechanical interactions to better understand the implanted joint integrity and the tissue quality at the hemi implant interface. The aim is to enhance patients’
quality of life, avoid should hemi-implant failure and revision.
Declarations
Conflict of interest: Amira Hanoun and Michel Hassler are employed by Tornier SAS that commercialize the pyrocarbon hemi
implant investigated in the current study. These authors, their immediate family, and any research foundation with which they are
affiliated did not receive any financial payments or other benefits from any commercial entity related to the subject of this article.
The other authors declare no conflict of interest.
Acknowledgements: This study was funded by Tornier SAS.
The authors wish to thank Dr. Olivier Menouillard (Ramsay
Santé, Höpital privé Pays de Savoie, Annemasse, France), Dr. Arnaud Godenèche (Hôpital privé Jean Mermoz Ramsay-GDS, Centre
Orthopédique Santy, Lyon, France), and Dr Robert Hudek (Klinik
für Schulter- und Ellbogenchirurgie, RHÖN KlinikumAG, Bad Neustadt, Germany) for the tissue samples and the clinical data about
the three cases investigated in the current study. The authors wish
to thank Imbert de Gaudemaris and Ghassen Ouenzerfi for their
contribution in the samples collection. The authors wish to thank
the lipidomic platform of IMBL (Institut Multidisciplinaire de Biochimie des Lipides) for the lipidomic analyses. The authors wish to
thank Novotech and CIQLE for their contribution in the histological preparations and analyses.
References
- Matsen FA, Clinton J, Lynch J, Bertelsen A, Richardson ML. Glenoid
component failure in total shoulder arthroplasty. J. Bone Jt. Surg.
2008; 90: 885-896.
- Papadonikolakis A, Neradilek MB, Matsen FA. Failure of the Glenoid Component in Anatomic Total Shoulder Arthroplasty. J. Bone Jt. Surg. 2013; 95: 2205-2212.
- Levine WN, Fischer CR, Nguyen D, Flatow EL, Ahmad CS, Bigliani
LU. Long-Term Follow-up of Shoulder Hemiarthroplasty for Glenohumeral Osteoarthritis. J. Bone Jt. Surg. 2012; 94: e164-1-7.
- Chan SMT, Neu CP, Komvopoulos K, Reddi AH, Di Cesare PE. Friction and Wear of Hemiarthroplasty Biomaterials in Reciprocating
Sliding Contact With Articular Cartilage. J. Tribol. 2011; 133.
- Klawitter JJ, Patton J, More R, Peter N, Podnos E, Ross M. In vitro
comparison of wear characteristics of PyroCarbon and metal on
bone: Shoulder hemiarthroplasty. Shoulder Elb. 2020; 12: 11-22.
- Vanlommel J, De Corte R, Luyckx JP, Anderson M, Labey L, Bellemans J. Articulation of Native Cartilage Against Different Femoral
Component Materials. Oxidized Zirconium Damages Cartilage Less
Than Cobalt-Chrome. J. Arthroplasty. 2017; 32: 256-262.
- Franceschetti E, Gregori P, Giurazza G, Papalia G, Caraffa A, Papalia
R. Short to Early-Mid Term Clinical Outcomes and Survival of PyrocarbonShoulder Implants: A Systematic Review and Meta-Analysis. J. Shoulder Elb. Arthroplast. 2023; 7: 247154922311521.
- Garret J, Harly E, Le Huec JC, Brunner U, Rotini R. Godenèche A.
Pyrolytic carbon humeral head in hemi-shoulder arthroplasty: preliminary results at 2-year follow-up. JSES Open Access. 2019; 3:
37-42.
- Impergre A, Trunfio-Sfarghiu AM, Wimmer MA. Evaluation of articular cartilage wear against pyrolytic carbon in the context of
spherical interposition shoulder arthroplasty. Biotribology. 2023;
33-34: 100237.
- McBride AP, Ross M, Hoy G, Duke P, Page R, Peng Y, Taylor F.
Mid-term outcomes of pyrocarbon humeral resurfacing hemiarthroplasty compared to metal humeral resurfacing and metal
stemmed hemiarthroplasty for osteoarthritis in young patients:
Analysis from the Australian Orthopaedic Association National
Joint Replace. J. Shoulder Elb. Surg. 2021.
- Coughlin TR, Kennedy OD. The role of subchondral bone damage
in post-traumatic osteoarthritis. Ann. N. Y. Acad. Sci. 2016; 1383:
58-66.
- Zarka M, Hay E, Ostertag A, Marty C, Chappard C, Oudet F, Engelke
K, Laredo JD, Cohen-Solal M. Microcracks in subchondral bone
plate is linked to less cartilage damage. Bone. 219; 123: 1-7.
- Elder SH, Cooley AJ, Borazjani A, Sowell BL, To H, Tran SC. Production of hyaline-like cartilage by bone marrow mesenchymal stem
cells in a self-assembly model. Tissue Eng. - Part A. 2019; 15; 3025-3036.
- Alliston T, Hernandez CJ, Findlay DM, Felson DT, Kennedy OD. Bone
marrow lesions in osteoarthritis: What lies beneath. J. Orthop.
Res. 2018; 36: 1818-1825.
- Martin R, Jakob RP. Review of K.H. Pridie (1959) on “A method of
resurfacing osteoarthritic knee joints.” J. ISAKOS. 2022; 7: 39-46.
- Broyles JE, O’Brien MA, Stagg MP. Microdrilling Surgery Augmented With Intra-articular Bone Marrow Aspirate Concentrate, Platelet-Rich Plasma, and Hyaluronic Acid: A Technique for Cartilage
Repair in the Knee. Arthrosc. Tech. 2017; 6: e201-e206.
- Hills BA. Surface-active phospholipid: A Pandora’s box of clinical
applications. Part II. Barrier and lubricating properties. Intern.
Med. J. 2002; 32: 242-251.
- During A, Penel G, Hardouin P. Understanding the local actions of
lipids in bone physiology. Prog. Lipid Res. 2015.
- Bartl R. Histology of Normal Bone and Bone Marrow, and Their
Main Disorders. 2013; 3-20.
- Lotz MK, Otsuki S, Grogan SP, Sah R, Terkeltaub R, D’Lima D. Cartilage cell clusters. Arthritis Rheum. 2010; 62: 2206-2218.
- Lotz MK, Loeser RF. Effects of aging on articular cartilage homeostasis. Bone. 2012; 51: 241-248.
- Benjamin M, Evans EJ. Fibrocartilage. J. Anat. 1990; 171: 1-15.
- Sophia Fox AJ, Bedi A, Rodeo SA. The Basic Science of Articular
Cartilage: Structure, Composition, and Function. Sport. Heal. A
Multidiscip. Approach. 2009; 1: 461-468.
- Sakai N, Hashimoto C, Yarimitsu S, Sawae Y, Komori M, Murakami
T. A functional effect of the superficial mechanical properties of
articular cartilage as a load bearing system in a sliding condition.
Biosurface and Biotribology. 2016; 2: 26-39.
- Bogoch ER, Lee TC, Fornasier VL, Berger SA. Articular damage is associated with intraosseous inflammation in the subchondral bone
marrow of joints affected by experimental inflammatory arthritis
and is modified by zoledronate treatment. J. Rheumatol. 2007; 34:
1229-1240.
- Li Z, Yao SJ, Alini M, Stoddart M. Chondrogenesis of human bone
marrow mesenchymal stem cells is modulated by frequency and
amplitude of dynamic compression and shear stress. Eur. Cells Mater. 2009; 18: 51.