Introduction
Ovarian Cancer (OC) is one of the common gynecological malignant tumors, and epithelial ovarian cancer is the main cause of
death [1]. Ovarian cancer accounts for about 4% of cancer morbidity and mortality among women worldwide, and the 5-year
survival rate in most countries is only about 30% to 40% [2].
Lymph node status is listed as a crucial variable in the Federation
International of Obstetrics and Gynecology (FIGO) ovarian cancer
staging system [3]. OC patients with lymph node metastasis are
divided into stage III or higher. Up to date, the traditional idea of
surgical treatment of ovarian cancer is to achieve complete Cytoreductive Surgery (CRS), while removing enlarged or suspected
lymph nodes [4]. However, there are still doubts about the need
for lymph node resection in early stage I-II patients, patients who
need to retain reproductive function, patients with low-grade serous cancer and patients with mucinous cancer, and for patients
with advanced OC, the effect of retroperitoneal lymphadenectomy on its prognosis is still controversial [4,5]. The results of a prospective controlled study in 2019 showed that for patients who
achieved complete CRS and had no enlarged lymph nodes before
or during operation, pelvic and para-aortic lymphadenectomy not
only did not achieve no survival benefit, but increased the risk of
postoperative complications [6]. Therefore, it is obviously unwise
to perform lymph node dissection on all patients with resectable
ovarian cancer, nor is it in line with the current era of individualized cancer medicine.
Enhanced MSCT is one of the routine methods to evaluate the
condition of patients with epithelial ovarian cancer before treatment. MSCT has superior advantages, including wide availability,
good repeatability, and fast image scanning time [7]. However, the
traditional interpretation of imaging images mostly depends on
the professional knowledge of imaging doctors and the experience of imaging diagnosis. There may be great differences in the
interpretation results of the same imaging data and different
imaging doctors. In the process of traditional imaging interpretation, it is often difficult to interpret the metastatic lymph nodes
of ovarian cancer due to the influence of pelvic mass and pelvic
and abdominal metastatic foci. Inexperienced doctors, especially
young imaging doctors are prone to misjudgment and omission.
At present, as a developing new research field, radiomics aims to
extract more complex feature information from traditional medical images, such as Computed Tomography (CT) and Magnetic
Resonance Imaging (MRI). These high-throughput objective image assembly features are extracted from the segmented ROI to
achieve the transformation from images to quantitative data established clinical models, including diagnosis, prognosis and prediction models [7-11].
At present, in the field of radiomics, there are many related
studies to predict the lymph node status of malignant tumors. In
recent years, in the related research of pelvic tumors, there are
many literatures about predicting lymph node metastasis of rectal cancer [12], cervical cancer [11] and endometrial cancer [13]
based on radiomics imaging characteristics, and the diagnostic efficiency turns out to be good.
There have been several previous studies based on CT texture
and neural network to predict the lymph node status of ovarian
cancer. In 2019, some scholars studied the diagnostic value of
LNM of ovarian cancer based on the lymph node texture analysis
of multi-slice spiral CT images of ovarian cancer patients. Finally,
the area under ROC of CT texture entropy was 0.83. However, it
only analyzed the texture features of a single CT rather than a
complete imaging analysis, without independent verification set
verification [14]. In 2020, Liu et al [15] studied the clinical value
of constructing pelvic and abdominal LNM assistant diagnosis
system for ovarian malignant tumors based on depth neural network in enhanced CT images, and its AUC is 0.7664. But no related
studies have been found to predict the LNM of ovarian cancer
founded on complete radiomics.
The aim of our study is to construct a pretreatment prediction
model of MSCT for epithelial OC founded on the imaging characteristics of preoperative LNM enhancement imaging in patients
with epithelial ovarian cancer, so as to improve the accuracy of
imaging evaluation of epithelial ovarian cancer before operation,
and then assist clinical selection of appropriate treatment strategies, including the choice of adjuvant treatment, the feasibility of
preserving fertility during surgical treatment, the determination
of surgical scope and so on.
Materials and methods
Research object
Patients with epithelial ovarian cancer confirmed by operation
and pathology in the first affiliated Hospital of Guangxi Medical
University from January 2015 to February 2021 were retrospectively analyzed. The review require approval by the Ethical Committee of First Affiliated Hospital of Guangxi Medical University.
Inclusion criteria: (1) patients was proved to be epithelial OC
by operation and pathology, and there were pathological results
of lymph node dissection, which included at least pelvic lymph
nodes and para-aortic lymph nodes; (2) plain and enhanced MSCT
scans of abdomen and pelvis were performed within 2 weeks before operation, and complete CT enhanced images were obtained,
and the image quality met the requirements of post-processing;
(3) there was no lack of clinical data and pathological information.
Exclusion criteria: (1) patients received preoperative radiotherapy, chemotherapy or other treatment, (2) patients with
distant metastasis or complicated with other primary malignant
tumors.
Information collection of patients enrolled in the group
(1) General data: Age, family history of tumor, menopausal
history.
(2) Laboratory examination parameters: Serum CA125
level and CA199 level within one week before operation, for serum CA125 level, record its specific value, unit is U/ml; for serum
CA199 level, record “0” = CA199 level is normal (<37 U/ml), “1” =
CA199 level is increased (≥ 37 U/ml).
(3) Pathological results: All pathological results were reviewed and signed by two pathologists with more than 8 years
experience in pathological diagnosis. Patients with multiple pathological results were subject to the last results or the pathological
results of in-hospital consultation. According to the pathological
results as the gold standard, if all the dissected lymph nodes of
the patient did not show LNM, the patient was recorded as “0” =
LNM negative; if one of the lymph nodes or more than two lymph
nodes were metastasized, the patient was recorded as “1” = LNM
positive.
Methods CT examination methods
All the patients in this study underwent multi-slice spiral CT
plain scan and enhanced examination within 2 weeks before operation, and all the patients signed the examination consent form
before the examination. During the examination, the patients
were instructed to take the supine position and use a high-pressure syringe to inject the nonionic contrast agent iohexol 300 ng/
ml (2.5-3.0 ml/kg) through the elbow vein with a dose of 3.0-3.5
mL/s. The contrast-enhanced MSCT images were obtained 60-80
seconds after injection of contrast medium. Scanning parameters:
scan layer thickness and interval is 1.0 mm~2.5 mm, matrix is 512
x 512, tube voltage is 120 KV, and the tube current is automatically adjusted according to the patient’s body shape (between
220~400 mAs). CT imaging equipment and scanning parameters
are shown in Table 1.
Table 1: Inspection equipment and scanning parameters.
| CT imaging device |
Thickness |
Tube voltage |
Matrix |
| Siemens Force CT |
1.0 |
120 |
512×512
|
| GE Revolution CT |
1.0 |
120 |
512×512
|
| Siemens Sensation64 |
2.0 |
120 |
512×512
|
| Siemens Dual Source |
1.25 |
120 |
512×512
|
| GE Light Speed VCT |
2.5 |
120 |
512×512
|
Note: All CT scanning devices are multi-layer spiral CT; tube current automatically adjusted according to the body type of the patient.
Some studies have shown that there is no significant difference
in the diagnostic efficiency of imaging modeling by selecting CT
images with different thickness for ROI sketching [16]. More
scholars obtain image data from multicenter institutions to
analyze imaging characteristics for retrospective study [17-21].
Image and data processing methods
Analysis of qualitative and quantitative imaging data
The films were read by a radiologist with 3 years' experience
and 5 years' experience in abdominal imaging diagnosis without
knowing the results of operation and pathology. When there is
a difference of opinion, discuss with another chief physician of
abdominal imaging diagnosis to get the final interpretation result.
When the lesions are bilateral ovarian lesions, the evaluation of
the following [2] is based only on the largest lesions. Observe the
following image features:
(1) Imaging evaluation of the location of the lesions,
unilateral or bilateral ovarian disease, "0" = unilateral ovarian
disease, "1" = bilateral ovarian disease.
(2) The two physicians evaluated the size of the tumor in
the axial and sagittal plane, and after reaching agreement, they
chose the same imaging orientation to measure, and calculated
the average value of the maximum length and diameter of the
tumor measured by the two physicians in the venous phase, from
the outer edge of the lesion to the outer edge (including the
capsule), avoiding the thickened blood vessels at the edge of the
lesion, including the whole lesions, in cm, keeping one digit after
the decimal point.
(3) With or without ascites, the presence or absence of
ascites in the abdominal and pelvic cavity was observed in CT
images. if there is a water-like density shadow in the abdominal
and pelvic cavity, no matter how much it is, record "1" = ascites,
and if there is no water-like density shadow in the abdominal and
pelvic cavity, it is recorded as "0" = no ascites.
(4) The lymph node status was evaluated by CT. The enlarged
lymph nodes in the abdominal and pelvic cavity were observed
in the arterial phase and venous phase of CT enhancement. If
the short diameter of the lymph node was ≥1 cm, liquefaction
and necrosis were seen in the lymph nodes, and "1" = positive
lymph node metastasis was recorded when the edge was circular
enhancement, and "0" = negative lymph node metastasis was
recorded if the short diameter <1 cm or fat and calcification in the
lymph nodes.
Image segmentation
The venous phase enhanced CT images (DICOM) of each
patient were downloaded from PACS and transmitted to Huiying
big data artificial intelligence research platform of Huiyi. Without
knowing the pathological results, the above two physicians were
respectively on the venous phase MSCT enhanced axial thin slice
(slice thickness of 1.0-2.5 mm) images of each patient. Manually
delineate ROI of the primary lesion along the edge of the lesion on
each continuous plane (Figure 1), carefully avoiding the thickened
blood vessels, fat, and intestinal wall at the edge of the lesion.
A month later, the same physician reconfirmed and adjusted
the boundary of the lesions. The difference of the boundary of
ROI was decided by two people through consultation, and the
confusion was finally decided by reading the film with another
chief physician of abdominal imaging diagnosis.
Feature extraction of image assemblage
Due to the image from different CT imaging devices, there are
different scanning parameters, the Radcloud platform is used to
pre-process the image before extracting the image features, so as
to reduce the influence of different image parameters on the extraction of image features. Then a total of 1409 imaging features
were automatically extracted from the segmented ROI on the
Radcloud platform and were divided into three portions. The first
portion (first-order statistical features) consists of 126 descriptors,
which quantitatively describe the distribution of voxel intensity
in MSCT images through common and basic metrics. The second portion (3D features) contains 14 3D features that reflect the
shape and size of the region. The third portion (texture features),
calculated from the gray running length and gray co-occurrence
texture matrix, includes 525 texture features that can quantify regional heterogeneity.
Feature selection and model construction
The redundant features were successively reduced by the variance threshold method and Select K Best method, and the LASSO
algorithm was processed to opt the optimal image features. For
the frontmost method threshold is 0.8, so that the variance eigenvalues less than 0.8 are removed. As for Select K Best method
threshold is 0.05 (P value), the features greater than 0.05 are removed. The cost function of LASSO model is L1 regularizer, which
set 5 as the error value of cross-validation, and 1000 as the maximum number of iterations. Based on the selected characteristics
of image group, this study uses support vector machine (SVM) to
construct the prediction model of image group through machine
learning. Then, the verification set and training set were randomly
divided in term of the proportion of 2:8. After statistical analysis,
the clinical data and CT imaging qualitative and quantitative data
were selected, and the imaging parameters were integrated to
construct a combined prediction model of lymph node metastasis
of epithelial ovarian cancer. The validity of the model was verified
in the internal independent test set. The diagnostic value of the
model was evaluated by the area under the working characteristic curve (AUC), sensitivity, specificity and 95% CI. Then, verify its
diagnostic performance in an independent test set.
Statistical analysis method
SPSS25.0 was used to analyze the data. Firstly, we performed
the ShapiroWilk test to test the data normality. The unit of measurement satisfies the normal distribution, and the independent
sample T test is used for univariate analysis. The measurement
data do not meet the normality, and the comparison of different
groups is expressed by nonparametric Mann-Whitney U rank sum
test in the form of M (P25, P75). Chi-square test or Fisher exact
test was used to compare the counting data, which was expressed
in the form of n (%). The best critical value of serum CA125 level
for predicting lymph node metastasis was obtained by ROC curve
and Jordan index. The variables with significant differences were
included in the multivariate analysis, and the multivariate binary
Logistic regression analysis was used to determine the independent predictors. Delong test was performed in MedCalc software
to compare the diagnostic efficiency of ROC curves of different
models. All the statistically significant data turn out to be P value
less than 0.05.
Results
Analysis of baseline data of patients in the group
Comparison of baseline data between training set and test
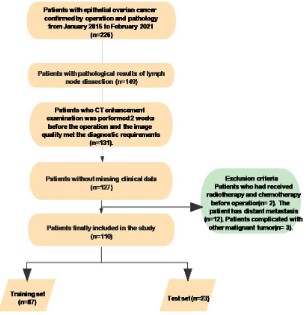
set: A total of 110 patients with epithelial OC were enrolled according to the enrollment conditions (Figure 2). The average age
was 48 ± 10.32, ranging from 23 to 70 years old.
Table 2: Comparison of baseline data between training set and test set.
|
|
Cases divided |
|
|
|
Variables |
group |
P value |
|
|
|
Training (n=87) |
test(n=23) |
|
| Age |
<45 |
31(83.7%) |
6(16.3%) |
0.389 |
|
≥45 |
56(76.7%) |
17(23.3%) |
|
| Family history of cancer |
yes |
4(57.2%) |
3(42.8%) |
0.14 |
|
no |
83(80.6%) |
20(19.4%) |
|
| Menopause |
yes |
35(71.4%) |
14(28.6%) |
0.077 |
|
no |
52(85.3%) |
9(14.7%) |
|
| CA125 (U/ml) |
≤297.8# |
54(79.4%) |
14(20.6%) |
0.916 |
|
>297.8# |
33(78.5%) |
9(21.5%) |
|
| CA199 (U/ml) |
<37 |
69(79.3%) |
18(20.7%) |
0.912 |
|
≥37 |
18(78.2%) |
5(21.8%) |
|
| Lesion location |
unilateral |
67(77.9%) |
19(22.1%) |
0.563 |
|
bilateral |
20(83.3%) |
4(16.7%) |
|
| Ascites |
yes |
53(81.6%) |
12(18.4%) |
0.448 |
|
no |
34(75.6%) |
11(24.4%) |
|
| Size (cm) |
<15 |
63(78.7%) |
17(23.3%) |
0.886 |
|
≥15 |
24(80.0%) |
6(20.0%) |
|
| CT to evaluate LNM |
Poc. |
69(78.4%) |
19(21.6%) |
0.725 |
|
Neg. |
18(81.8%) |
4(18.2%) |
|
| LNM pathology |
Neg |
60(81.2%) |
16(18.8%) |
0.956 |
|
Neg |
27(79.4%) |
7(20.6%) |
|
Note: #indicates that 297.8 U/ml is the best critical value of CA125 level
for predicting lymph node metastasis verified by ROC curve and Jordan
index. n is the number of cases.
Among the 110 patients with epithelial ovarian cancer, 34 cases were pathologically confirmed to be LNM positive, accounting
for 30.9% of 76 cases with negative LNM, accounting for 69.13%.
The training set (n=87) and test.
set (n=23) of OC patients were randomly divided in term of the
proportion of 8:2. The constituent ratio of patient baseline data
between training set and verification set turn out to be no significant difference (Table 2).
Results of univariate analysis
The results showed that the P value of lesion location, CA125
level and CT evaluation of lymph node status in lymph node
metastasis were all less than 0.05, but there were no significant
differences in age, family history of cancer, menopause, preoperative CA199 level, ascites and maximum
line diameter (P>0.05). The sensitivity of CT in the assessment of
lymph node metastasis was 41.2% (14/34) and the specificity was
89.4% (68/76).
Table 3: Results of univariate analysis.
|
|
LNM |
|
|
|
| Variables |
groups |
Neg. (n=76) |
Poc. (n=34) |
P values |
|
| Age |
<45 岁 |
29(78.3%) |
8(21.7%) |
0.133 |
|
| ≥45 岁 |
47(64.3%) |
26(35.7%) |
|
|
| Family history
of cancer |
yes |
5(71.4%) |
2(28.6%) |
0.89 |
|
| no |
71(69.0%) |
32(31.0%)
|
|
|
| Menopause |
yes |
32(66.7%) |
17(33.3%) |
0.441 |
|
| no |
44(71.2%) |
17(28.8%)
|
|
|
| CA125 (U/ml) |
≤297.8 |
55(78.9%) |
13(21.1%) |
0.001 |
|
| >297.8 |
21(50.0%) |
21(50.0%) |
|
|
| CA199 (U/ml)
|
<37 |
60(61.5%) |
27(38.5%) |
0.956 |
|
| ≥37 |
16(87.6%) |
7(10.4%) |
|
|
| Lesion location |
unilateral |
65(74.7%) |
21(25.3%) |
0.005 |
|
| bilateral |
11(45.0%) |
13(55.0%) |
|
|
| Ascites |
no |
31(68.9%) |
14(31.1%) |
0.97 |
|
| yes |
45(69.3%) |
20(30.7%) |
|
|
| Size(cm) |
<15cm |
52(65.0%) |
28(35.0%) |
0.129 |
|
|
≥15cm |
24(80.0%) |
6(20.0%) |
|
| CT to
evaluate
LNM |
Neg. |
68(77.3%) |
20(22.7%) |
<0.001 |
| Poc. |
8(36.3%) |
14(63.7%) |
|
Note: If there is no special note, all the results are statistically
significant (P<0.05).
Results of multivariate Logistic regression analysis
The variables with statistically significant differences in lymph
node metastasis (lesions location, CA125 level, CT evaluation of
lymph node status) were included in multivariate Logistic regression analysis, and multivariate analysis was carried out. Multivariate analysis using binary Logistic regression analysis showed
that CA125 level (P<0.05), CT evaluation of lymph node status
(P<0.05) and lesion location (P<0.05) were independent predictors of lymph node metastasis in patients with ovarian cancer, as
shown in Table 4.
Table 4: Multivariate Logistic regression analysis results.
| Variables |
B |
SE |
Wald |
P |
OR* |
95% CI |
| CA125 |
1.031 |
0.475 |
4.722 |
0.03 |
2.804 |
1.106-7.109 |
| CT to evaluate LNM |
0.936 |
0.54 |
8.404 |
0.004 |
5.022 |
1.687-14.954 |
| Lesions location |
1.174 |
0.557 |
4.731 |
0.03 |
3.234 |
1.123-9.109 |
| Constant |
-1.92 |
0.367 |
27.449 |
<0.001 |
0.147 |
|
Note: * Indicates the number of contrast, that is, a multiple concept
index, with serum CA125 level ("0", that is, ≤ 297.8U/ml), CT evaluation
of lymph node status ("0", that is, lymph node metastasis negative) and
lesions location ("0", that is, unilateral) as control items.
Construction of radiomics model
Feature extraction and radiomics model construction: The
dimensionality reduction analysis and machine learning of
radiomics are carried out on the Radcloud platform. Firstly, the
variance threshold method is used to select 398 features from
1409 features, and then 90 features are selected by select Kbest
method. Finally, five optimal imaging features (Table 5, Figure 3)
are selected by LASSO algorithm, which are represented by R1-R5,
including three gray correlation matrix features (R1-R3) and two
first-order statistical features (R4, R5).
Table 5: Lasso coefficient of image omics eigenvalues.
| Radiomic feature |
Radiomic class
Filter |
P value Lasso |
|
|
| R1: Dependence Variance |
gldm |
wavelet-HHL |
<0.001 |
0.05802 |
| R2: Dependence Variance |
gldm |
wavelet-LHH |
<0.001 |
0.05513 |
| R3: Large Dependence Low |
gldm |
wavelet-LHH |
0.005 |
-0.03292 |
| Gray Level Emphasis |
| R4: Skewness |
first order |
wavelet-LLL |
0.002 |
-0.00175 |
| R5: Skewness |
first order |
square |
0.002 |
-0.05067 |
Notes: gldm: gray level dependence matrix; DV: Dependence Variance;
LDLGLE: Large Dependence Low Gray Level Emphasis; Lasso coefficient:
The negative value of indicates that there is a negative correlation
between the feature and lymph node metastasis, while a positive value
indicates that the feature is positively correlated with lymph node
metastasis. The greater the absolute value, the greater the correlation.
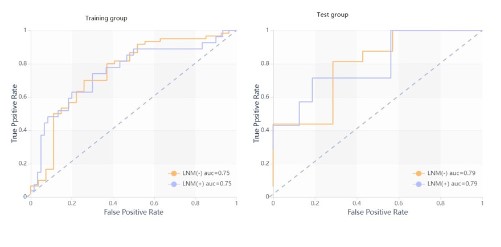
Table 6: Diagnostic efficiency of the radiomics model.
| Group |
AUC |
95% CI |
Sensitivity |
Specificity |
| Training set |
0.754 |
0.65-0.86 |
0.74 |
0.7 |
| Test set |
0.795 |
0.65-0.86 |
0.71 |
0.69 |
Note: The results of ROC curve of training set and test set of radiomics
model.
Evaluation and verification of radiomics model.
ROC curve analysis results of radiomics model are shown in
Table 6 and Figure 4.
Construction of combined model
Feature extraction and selection: On the Radcloud platform
finally select the five optimal imaging features (R1~R5), which are
the same as the imaging model, as shown in Table 5 and Figure 3.
Five imaging features selected from the statistically significant baseline data (CA125 level, lesions location, CT evaluation of
lymph node status) were combined with SVM classifier to build a
combined model, and the SVM parameters were consistent with
the radiomics model parameters.
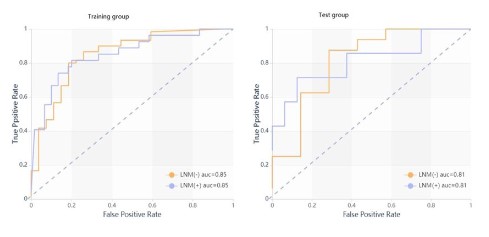
Evaluation and verification of combined model
ROC curve analysis results of combined model are shown in
Table 7 and Figure 5.
Table 7: Combined model of clinical data, CT signs and imaging
omics features.
| Group |
AUC |
95% CI |
Sensitivity |
Specificity |
| Training |
0.849 |
0.76- |
0.81 |
0.8 |
| Test set |
0.813 |
0.76- |
0.71 |
0.81 |
Note: The AUC value of combined model training set and test set is 0.85
and 0.81 respectively.
Comparison of prediction efficiency of different models.
The Delong test of the performance of the two models predicting LNMROC is shown in Table 8. The results showed that the AUC
of the combined model for predicting ovarian cancer LNM in the
training set and verification set was higher than that of the single
radiomics model. There was a significant difference in the prediction efficiency of the two models in the training set (Table 8 and
Figure 6).
Discussion
From now on, there are different methods to predict lymph
node metastasis of tumor patients through imaging models.
Based on different imaging images (CT, MRI, US, PET/CT, etc.), primary tumor, primary tumor and peritumoral structure, primary
tumor and surrounding lymph nodes, regional lymph nodes are
segmented as ROI, and then imaging features are extracted to
establish LNM prediction models. There are a variety of feature selection and modeling methods. However, there are few studies
based on MSCT imaging in the field of ovarian cancer, and there
are even fewer studies on the application of imaging model to
predict the LNM of ovarian cancer. This study is based on MSCT
imaging, using LASSO algorithm to select the best imaging features of LNM in MSCT, combined with traditional data related to
ovarian cancer LNM, to explore the value of the model in predicting LNM of epithelial ovarian cancer.
Table 8: Delong test results of ROC curve efficiency of both models.
|
Models |
AUC |
SE |
95% CI |
P value |
| Training set |
Radiomics model |
0.754 |
0.060 |
0.65-0.86 |
0.023 |
|
Combined model |
0.849 |
0.047 |
0.76-0.94 |
|
| Test set |
Radiomics model |
0.795 |
0.109 |
0.65-0.86 |
0.719 |
|
Combined model |
0.813 |
0.114 |
0.76-0.94 |
|
Note: The prediction efficiency of ROC curve of the two models was
compared by Delong test, P < 0.05. The difference was statistically
significant.
Analysis of baseline data of enrolled
According to the relevant literature, the positive rate of LNM
in ovarian cancer is about 44% to 60%, and the metastasis rate of
patients with advanced ovarian cancer can be as high as 40% to
73.7%. The probability of LNM is also different due to different
clinical stages, histopathological types, and degree of differentiation, while serous and clear cell carcinoma have a higher risk of
LNM than other histological types of ovarian cancer [19,22-24].
In this study, there were 34 cases of LNM positive group (30.9%)
and 76 cases of negative lymph node metastasis group (69.13%).
Among the 87 cases in the training set, 27 cases were pathologically confirmed lymph node metastasis (31.0%), and 7 cases in the
test set 23 cases (the metastasis rate was 30.4%). In this study,
the results showed that the preoperative CA125 level of ovarian
cancer patients with LNM positive was higher. This study showed
that the rate of lymph node metastasis increased in serum CA125
>297.8 U/ml group, and the probability of lymph node metastasis
in CA125 ≤ 297.8 U/ml group was 2.804 times higher than that
in CA125 ≤ 297.8 U/ml group. Although the critical value is different from that of previous studies (the critical value of CA125
is 535 U/ml) [25], the results show that the level of CA125 is an
independent risk factor for evaluating LNM in ovarian cancer. The location of the lesions was an important risk factor for evaluating
LNM in OC. The portion of lymph node metastasis in patients with
bilateral OC was significantly higher than that in patients with unilateral (left or right) lesions, which was consistent with the results
of other studies [14,26-28]. As a qualitative radiological feature,
preoperative CT evaluation of lymph node status can be easily obtained. This study shows that CT evaluation of lymph node status
is an independent risk factor for LNM.
However, our study showed that the age, family history of
tumor, menopause, preoperative CA199 level, maximum tumor
diameter and LNM in patients with OC with or without ascites
are not independent variables. It is worth noting that the level of
CA199 is an independent risk factor for LNM in patients with OC
in previous research. Early diagnosis of retroperitoneal LNM of
OC could be achieved by a combination of serum CA125, VEFG-
C and CA199 levels, which has clinical predictive value [29]. On
the basis of the univariate correlation between CA199 level and
LNM in this study, it does not show sufficient predictive strength
of CA199, which makes the exclusion of this variable a factor in
the construction of a combined model. However, the rejection of
important predictors may be the result of sample size, nuances in
data sets, or be confused by other predictors. For these predictors, the non-significant statistical association with lymph node
metastasis does not necessarily mean that CA199 levels are not
important. The insignificance of other characteristics may be due
to insufficient sample size or confusion among predictive factors,
which need to be confirmed by further study.
Construction of radiomics model
Traditional human eye film reading is usually limited by a variety of subjective factors, but the emerging radiomics (Radiomics)
breaks through these limitations to achieve the transformation
from image to data, so as to obtain more information, and then
improve the diagnostic efficiency. Its fast, non-invasive, low cost,
high patient acceptance and other advantages promote its rapid
development. As more and more variables are collected, high-dimensional data attract more and more attention in images. SVM,
as a Machine Learning (ML) tool to deal with classification problems, has a good performance in the classification and prediction
of high throughput data, even in ML tasks with limited samples
[30].
In recent years, radiomics has a good performance in the classification and prediction of OC. In this study, based on MSCT images, the LNM predictive assembly model of epithelial ovarian cancer was constructed on the SVM classifier. The ROC results show
that it has a certain diagnostic efficiency. Yu et al [14]. Studied
the diagnostic value of metastatic lymph nodes of ovarian cancer
based on multi-slice spiral CT image texture analysis, but only a
single CT texture feature analysis rather than a complete imaging
analysis, without independent verification set verification.
Combined model construction
In this study, univariate and multivariate Logistic regression
analysis of 9 LNM-related clinical data and CT signs (age, family
history of tumor, menopause, CA125, CA199, lesion location, tumor maximum diameter, ascites, and lymph node status assessed
by CT) showed that three factors (CA125 level, lesion location and
CT evaluation of lymph node status) were independent predictors of LNM in epithelial ovarian cancer. Combined with the selected
five optimal imaging features, a combined prediction model is
constructed. The combined model achieves good diagnostic efficiency, and the AUC of training set and test set are 0.849 and
0.813, respectively.
Some scholars have studied the preoperative serum CA125
level to predict the LNM of epithelial OC. The results showed that
the ROC curve showed the best critical value (535 U/ml) of pre-operative serum CA125 level, with a sensitivity and specificity of
70.0% and 83.1%, respectively. Imaging studies combined with
preoperative serum CA125 levels showed the highest sensitivity (90.0%). Imaging studies alone showed the highest specificity
for predicting lymph node metastasis (89.8%) [25]. It shows that
the diagnostic efficacy of single factor or single sign in predicting
LNM is lower than that of combining multiple factors and multiple
factors. However, this study only analyzes the predictive performance in terms of sensitivity and specificity, and does not use
other indicators to evaluate the predictive efficiency. This study
combines multiple clinical data, CT signs and imaging features to
construct a combined model. The AUC, specificity, sensitivity and
95% confidence interval are used to evaluate the prediction efficiency of the model, so as to improve and further increase the
stability and credibility of the model. In 2020, Liu et al [31] studied
the clinical value of the assistant diagnosis system of ovarian cancer pelvic and abdominal LNM based on depth neural network in
enhanced CT imaging. The ROC curve analysis results show that
the AUC is 0.7664, which is close to the diagnostic efficiency of the
imaging model, but lower than our combined model.
Comparison of prediction efficiency of different models
The disease assessment needs to integrate clinical, pathological, imaging and other different aspects of information. In this
study, it showed that the diagnostic efficiency of the combined
model is higher than that of the single one. Studies have shown
that the accuracy of CT in the diagnosis of pelvic lymph node and
retroperitoneal LNM in patients with OC is 48.15% (13/27) and
41.67% (10/24), respectively, and its accuracy is low [32]. In this
study, it turns out to be that simple CT signs have a certain rate of
missed diagnosis of lymph node metastasis in patients with ovarian cancer. Moreover, the combined model can predict the LNM
of patients with epithelial ovarian cancer with high sensitivity and
specificity.
This study has the following shortcomings as follows: (1) The
research object is a single center study, and the sample size is
small. (2) This study is a retrospective study, the model is only
verified in the internal independent test set, and there is no external verification and prospective verification. (3) There are few
LNM-related baseline data collected and analyzed in patients with
epithelial ovarian cancer. In addition to these characteristics analyzed in this study, epithelial ovarian cancer also has other risk factors for LNM. In addition, gene markers such as tumor suppressor
gene P16 [33] and BRCA mutation [34] were not considered in this
study. Further research is needed to explore the possibility of these
factors adding to the comprehensive prediction model of LNM.
Conclusion
This study confirmed the correlation between preoperative enhanced MSCT imaging parameters and LNM in patients with epithelial ovarian cancer, and established a preoperative combined
prediction model of LNM of epithelial ovarian cancer based on
MSCT imaging parameters and traditional baseline data (lesion
location and CA125 level (297.8 U/ml), CT evaluation of lymph
node status). The model achieved high diagnostic efficiency and
was verified in the internal independent test set. This model has
a certain value in improving the preoperative accuracy of ovarian
cancer and guiding clinical decision-making.
Declarations
Ethical approval and consent to participate: The review require approval by the Ethical Committee of First Affiliated Hospital
of Guangxi Medical University.
Consent for publication: Not application.
Funding: This study was granted from National Natural Science
Foundation of China (No. 81360220).
Competing interests: The authors have declared that no competing interest exists.
Author Contribution section: Jia-mei Li and Hua-li Xuan drafted and prepared manuscript for final publication; Jiamei Li and
Hua-li Xuan reviewed the literature and extracted the data; Jinyuan Liao worked on the statistic part; Jia-mei Li and Hua-li Xuan
drafted the manuscript; Jinyuan Liao performed consultation and
revised the manuscript. All authors issued final approval for the
version to be submitted.
Availability of supporting data: Not applicable.
Acknowledgements: Not applicable.
References
- F Bray, J Ferlay, I Soerjomataram, RL Siegel, LA Torre, et al. Global cancer statistics 2018: GLOBOCAN estimates of incidence and
mortality worldwide for 36 cancers in 185 countries. CA Cancer J
Clin. 2018; 68: 394-424.
- LA Torre, F Islami, RL Siegel, EM Ward, A Jemal. Global Cancer in
Women: Burden and Trends. Cancer Epidemiol Biomarkers Prev.
2017; 26: 444-457.
- J Prat. FIGO’s staging classification for cancer of the ovary, fallopian
tube, and peritoneum: Abridged republication. J Gynecol Oncol.
2015; 26: 87-9.
- L Huai-wu, X Miao-chun, Z Yu-hao, H Chun-ying, L Zhong-qiu. Interpretation of 2021 NCCN guidelines to the clinicalpratice in ovary
cancer including fallopian tubercancer and primary peritoneal
cancer (version1). Chinese Journal of Practical Gynecology and
Obstetrics. 2021; 37: 457-466.
- P Harter, J Sehouli, D Lorusso, A Reuss, I Vergote, et al. A Randomized Trial of Lymphadenectomy in Patients with Advanced Ovarian
Neoplasms. N Engl J Med. 2019; 380: 822-832.
- G Danala, T Thai, CC Gunderson, KM Moxley, K Moore, et al. Applying Quantitative CT Image Feature Analysis to Predict Response of
Ovarian Cancer Patients to Chemotherapy. Acad Radiol. 2017; 24:
1233-1239.
- RJ Gillies, PE. Kinahan, H Hricak. Radiomics: Images Are More than
Pictures, They Are Data. Radiology. 2016; 278: 563-77.
- ZY Shu, SJ Cui, X Wu, Y Xu, P Huang, et al. Predicting the progression of Parkinson’s disease using conventional MRI and machine
learning: An application of radiomic biomarkers in whole-brain
white matter. Magn Reson Med. 2021; 85: 1611-1624.
- N Kadoya, S Tanaka, T Kajikawa, S Tanabe, K Abe, et al. Homology-based radiomic features for prediction of the prognosis of lung
cancer based on CT-based radiomics. Med Phys. 2020; 47: 2197-2205.
- J Jian, Y Li, PJ Pickhardt, W Xia, Z He, et al. MR image-based radiomics to differentiate type Ι and type ΙΙ epithelial ovarian cancers. Eur Radiol. 2021; 31: 403-410.
- M Ciolina, V Vinci, L Villani, S Gigli, M Saldari, et al. Texture analysis versus conventional MRI prognostic factors in predicting tumor
response to neoadjuvant chemotherapy in patients with locally
advanced cancer of the uterine cervix. Radiol Med. 2019; 124:
955-964.
- YQ Huang, CH Liang, L He, J Tian, CS Liang, et al. Development and
Validation of a Radiomics Nomogram for Preoperative Prediction
of Lymph Node Metastasis in Colorectal Cancer. J Clin Oncol. 2016;
34: 2157-64.
- BC Yan, Y Li, FH Ma, GF Zhang, F Feng, et al. Radiologists with MRI-based radiomics aids to predict the pelvic lymph node metastasis
in endometrial cancer: A multicenter study. Eur Radiol. 2021; 31:
411-422.
- Y zhipeng, M Cunzhong, Z Changxiu, C Bin, Z Xiaofang, et al. Evaluation of lymph node metastasis in ovarian cancer based on CT
image texture analysis technique. Journal of Modern Oncology.
2020; 28: 3222-3226.
- L Wei, Z Dan, S Kejun, L teng, L Fei et al. Clinical application of
preoperative aided diagnosis system for metastatic lymph nodes
of pelvic and abdominal cavity based on deep neural network in
enhanced CT. Progress in Obstetrics and Gynecology. 2020; 29:
726-729+733.
- L Meng, D Dong, X Chen, M Fang, R Wang, et al. 2D and 3D CT
Radiomic Features Performance Comparison in Characterization of
Gastric Cancer: A Multi-Center Study. IEEE J Biomed Health Inform.
2021; 25: 755-763.
- L. R. F. Branco, R. B. Ger, D. S. Mackin, S. Zhou, L. E. Court and R.
R. Layman: Technical Note: Proof of concept for radiomics-based
quality assurance for computed tomography. J Appl Clin Med Phys.
2019; 20 199-205.
- MA Arshad, A Thornton, H Lu, H Tam, K Wallitt, et al. Discovery
of pre-therapy 2-deoxy-2-(18)F-fluoro-Dglucose positron emission tomography-based radiomics classifiers of survival outcome
in nonsmall-cell lung cancer patients. Eur J Nucl Med Mol Imaging.
2019; 46: 455-466.
- L Minig, F Heitz, D Cibula, JN Bakkum-Gamez, A Germanova, et
al. Patterns of Lymph Node Metastases in Apparent Stage I Low-Grade Epithelial Ovarian Cancer: A Multicenter Study. Ann Surg
Oncol. 2017; 24: 2720-2726.
- YX Ge, J Li, JQ Zhang, SF Duan, YK Liu et al. Radiomics analysis of
multicenter CT images for discriminating mucinous adenocarcinoma from nomucinous adenocarcinoma in rectal cancer and comparison with conventional CT values. J Xray Sci Technol. 2020; 28:
285-297.
- GW Ji, FP Zhu, Q Xu, K Wang, MY Wu, et al. Radiomic Features at
Contrast-enhanced CT Predict Recurrence in Early Stage Hepato-cellular Carcinoma: A Multi-Institutional Study. Radiology. 2020; 294: 568-579.
- J Zhou, JY Sun, SG Wu, X Wang, ZY He, et al. Risk factors for lymph
node metastasis in ovarian cancer: Implications for systematic
lymphadenectomy. Int J Surg. 2016; 29: 123-7.
- F Heitz, P Harter, B Ataseven, S Heikaus, S Schneider, et al. Stage-
and Histologic Subtype-Dependent Frequency of Lymph Node Metastases in Patients with Epithelial Ovarian Cancer Undergoing Systematic Pelvic and Paraaortic Lymphadenectomy. Ann Surg Oncol.
2018; 25: 2053-2059.
- O Ercelep, M Ozcelik, M Gumus. Association of lymphadenectomy
and survival in epithelial ovarian cancer. Curr Probl Cancer. 2019;
43: 151-159.
- HS Kim, NH Park, HH Chung, JW Kim, YS Song et al. Significance of
preoperative serum CA-125 levels in the prediction of lymph node
metastasis in epithelial ovarian cancer. Acta Obstet Gynecol Scand.
2008; 87: 1136-42.
- F. Xin-fei, M. Qing-song and L. Jun: Risk factors of retroperitoneal
lymph node metastasis of ovarian cancer and its image features of
MRI and CT. Chinese Journal of CT and MRI. 2020; 18: 134-138.
- T Mitamura, M Sekine, M Arai, Y Shibata, M Kato, et al. Risk factors
for lymph node metastasis of ovarian, fallopian tube and primary
peritoneal cancer in hereditary breast and ovarian cancer syndrome. Jpn J Clin Oncol. 2020; 50: 1380-1385.
- J Yang, J Ma, S Cheng, Y Wang. The Combination of Plasma Fibrinogen Concentration and Neutrophil Lymphocyte Ratio (F-NLR) as a
Prognostic Factor of Epithelial Ovarian Cancer. Onco Targets Ther.
2020; 13: 7283-7293.
- T Ying, Z Changxin, L Yongming, L Zongting. Value of combined detection of CA125 and VEGF-C, β2-MG in early diagnosis of ovarian
cancer lymph node metastasis. International Journal of Laboratory
Medicine. 2016; 37: 1930-1932.
- CY Guo, YC Chou. A novel machine learning strategy for model selections-Stepwise Support Vector Machine (StepSVM). PLoS One.
2020; 15: e0238384.
- H Kim, N Park, H Chung, J Kim, Y Song et al. Significance of preoperative serum CA-125 levels in the prediction of lymph node metastasis in epithelial ovarian cancer. 2008; 87: 1136-1142.
- T Bao-li, Y Ming, G Li-fu, L Xiao-ping, L Bing-yan, et al. Comparative study of the diagnosis value of MRI and CT in the pathological
stage, recurrence and metastasis of ovarian cancer. Progress in
Modern Biomedicine. 2020; 20: 4710-4713.
- H Wang, J Zheng, Q Li, M Zhou, D Ai, et al. Expression of the tumor
suppressor gene p16, and lymph node metastasis in patients with
ovarian cancer. Oncol Lett. 2017; 14: 4689-4693.
- A Meier, H Veeraraghavan, S Nougaret, Y Lakhman, R Sosa, et al.
Vargas: Association between CT-texture-derived tumor heterogeneity, outcomes, and BRCA mutation status in patients with high-grade serous ovarian cancer. Abdom Radiol (NY). 2019; 44: 2040-2047.