Introduction
Constrictive Pericarditis (CP) results from pericardial inflammation and fibrosis, impeding diastolic cardiac filling and eventually
leading to severe diastolic failure [1,2]. The accurate incidence
was not well established but estimated to be relatively rare, less
than no more than 1 per 100000 people per year [3]. There was
a regional heterogeneity regarding etiologies worldwide. Tuberculosis was the predominant cause of CP in developing countries,
while prior cardiac surgery was most frequent in North America
and Europe [4]. Additionally, other potential causes encompassed
asbestosis, trauma, malignancy, rheumatologic disease, and infection. A vast array of organisms has been demonstrated to be responsible for the development of CP such as human immunodeficiency virus, cytomegalovirus, adenoviruses, enteroviruses, and
influenzas. Despite the advance in diagnostic technique in terms
of immunohistochemistry, pericardioscopy, and polymerase chain
reaction testing, there were a significant proportion of patients
remains idiopathic [5]. Herein, we reported a CP patient associated with human herpesvirus 1 and 6B as determined by Next-Generation Sequencing (NGS).
Case presentation
A 37-year-old man was admitted to our hospital due to a six-month history of progressive exertional dyspnea and chest distress. He was previously diagnosed with a dual infection of hepatitis B and C and without tuberculosis one year ago. On physical
examination, a distention of jugular vein, a palpated hepatomegaly, and an auscultatory pericardial knock were identified. Cardiac
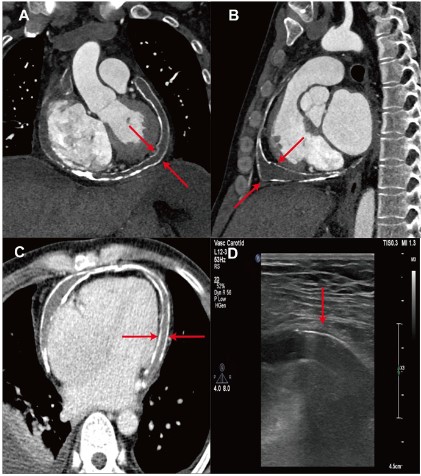
computed tomography confirmed the CP as a consequence of a
circumferential calcification of both the parietal and visceral layer
pericardium (Figure 1A-C). Transthoracic echocardiogram demonstrated the posterior motion of the ventricle septum at the
early-diastolic period in inspiration, a distention of inferior vena
cava with inspiratory collapse over 50%, and distinctly thickened
pericardium (Figure 1D), reconfirming the diagnosis. Abdominal
ultrasound revealed liver cirrhosis with multiple gallstones.
A radical pericardiectomy without cardiopulmonary bypass
establishment via median sternotomy was performed, showing a
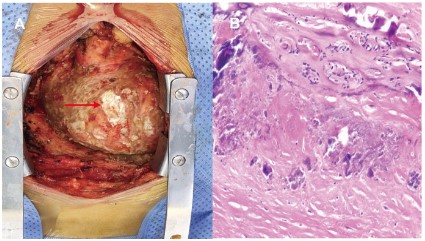
massive bean dreg-like substance wrapped between the parietal
and visceral layer pericardium. The calcific pericardium was removed as much as possible, inevitably leaving plaque-like epicardial calcification (Figure 2A). Care was taken not to injure the coronary vessels or the phrenic nerves. Pathology indicated significant
collagen hyperplasia, severe calcification, as well as aggregation
of a few neutrophils and lymphocytes (Figure 2B). What’s more,
human herpesvirus 1 and 6B rather than mycobacterium tuberculosis were detected as causes of NGS. The patient received diuretics and antiviral treatment postoperatively and was discharged
2 weeks after surgery with significantly relieved symptoms. The
postoperative one-month and three-month follow-ups showed a
satisfactory improvement in the New York Heart Association functional class, with 63% of left ventricle ejection fraction.
Discussion
The human herpesvirus identified as the cause of CP has been
rarely reported. This report described that a male with severe calcific CP confirmed by NGS underwent a meticulous radical pericardiectomy and showed a satisfactory outcome during follow-up.
The CP that represented a spectrum of diastolic heart failure
was relatively rare worldwide. It showed quite temporal and
regional differences with respect to the etiologies. Specifically,
there was a great etiology transition in the developed countries in
the 20th century, predominantly due to the widespread of cardiac
surgery [6]. It was reported that 0.2-0.4% of patients undergoing
cardiac surgery would result in CP after a postoperative mean of
2 years [7]. Of CP patients at the Mayo Clinic during the period
of 1936-1982, there was only 2% resulting from the prior cardical [8]. However, the incidence rose to 34% between 1996 and
2006 [9]. In contrast, tuberculosis was the most common cause in
developing regions such as Africa and Asia. For instance, the incidence of CP attributed to tuberculosis in India was 61% during the
period of 1954-1985 and 93% during the period of 1985-2004, respectively [9]. A recent systematic review including 30 studies and
over 11000 patients showed that tuberculosis remains the most
frequent cause in Asia and Africa while previous cardiac surgery
has become the predominant cause in North America and Europe
[4]. Regardless, a part of CP patients remained idiopathic. Nakanishi et al. described a CP patient associated with severe acute
respiratory syndrome coronavirus 2 vaccination [10]. Griessel et
al. first verified the Nocardia asiatica infection responsible for
the development of CP in a man concurrent with human immunodeficiency virus infection [11]. To the best of our knowledge,
we reported the first case confirming the human herpesvirus infection as the primary cause of CP with the utilization of NGS of
the pericardium sample. Additionally, the CP could be classified
into transient, effusive-constrictive, and calcific types. After sternotomy, the patient presented a severely calcific CP in view. The
calcific CP was reported to account for approximately 25%-30% of
all CP patients more prevalently radiation-induced and idiopathic
[12-14], which was different from that induced by human herpesvirus in our case.
The surgical pericardiectomy is the only curable intervention
for CP patients. Due to the severe pericardium calcification and
myocardial adherence, a meticulous pericardiectomy with or
without additional procedures such as the sacrifice of the phrenic
nerve, cardiopulmonary bypass establishment, ultrasonic decalcification, and careful wedge excision of calcific plaques, is required.
Anterior pericardiectomy was demonstrated to be inherent with
the residual calcific pericardium, a higher risk of recurrent CP,
thereby resulting in repeat pericardiectomy with 7% of 30-day
mortality [15]. Given this, we performed a complete radical pericardiectomy with the aim to remove the calcific pericardium as
much as possible. Postoperatively, the patient’s symptoms were
significantly relieved and treated with antivirus therapy. During
the 3-month follow-up, the patient showed a satisfactory outcome in terms of improvement in New York Heart Association
functional class and left ventricle ejection fraction.
Therefore, the human herpesvirus infection was first demonstrated to be capable of potentially triggering pericardial inflammation and fibrosis, thereby leading to CP. A complete radical
pericardiectomy to remove the pericardium as much as possible
was effective in such patients.
Declarations
Acknowledgments: We are grateful for the helpful comments
from each member of Zhuang’s and Chen’s groups.
Conflict of interest: There is no conflict of interest to declare.
Ethics Statement: This study was approved by the Guangdong
Provincial People’s Hospital ethics committee. Informed consent
was obtained from this patient.
Funding information: The present work was supported
by National Key Research and Development Program of China (No. 2020YFC1107904), Science and Technology Fundation for Guangzhou Health (2023A031004), Guangdong peak
project (DFJH201802; DFJH2020029), Science and Technology Planning Project of Guangdong Province (2019B020230003; 2018B090944002; 2020B1111170011), and Science and Technol-
ogy Program of Guangzhou (202206010049).
References
- Lin J, Li M, Huang Y, et al. Evaluation of Pericardial Thickening and
Adhesion using High Frequency Ultrasound. 2023; 3: S0894-7317:
00190-6.
- Izumi C, Iga K Fau - Sekiguchi K, Sekiguchi K Fau - Takahashi S, Takahashi S Fau - Konishi T, Konishi T. Usefulness of the transgastric
view by transesophageal echocardiography in evaluating thickened pericardium in patients with constrictive pericarditis.
- Mori M, Mullan CW, Bin Mahmood SU, et al. US National Trends in
the Management and Outcomes of Constrictive Pericarditis: 2005-2014. Can J Cardiol. 2019; 35: 1394-1399.
- Kosmopoulos M, Liatsou E, Theochari C, et al. Updates on the
Global Prevalence and Etiology of Constrictive Pericarditis: A Systematic Review. Cardiol Rev. 2023.
- Maisch B, Ristic AD. The classification of pericardial disease in the
age of modern medicine. Curr Cardiol Rep. 2002; 4: 13-21.
- Ling LH, Oh JK, Schaff HV, et al. Constrictive pericarditis in the modern era: Evolving clinical spectrum and impact on outcome after
pericardiectomy. Circulation. 1999; 100: 1380-6.
- Im E, Shim CY, Hong GR, et al. The incidence and clinical outcome
of constrictive physiology after coronary artery bypass graft surgery. J Am Coll Cardiol. 2013; 61: 2110-2.
- McCaughan BC, Schaff HV, Piehler JM, et al. Early and late results
of pericardiectomy for constrictive pericarditis. J Thorac Cardiovasc Surg. 1985; 89: 340-50.
- Syed FF, Schaff HV, Oh JK. Constrictive pericarditis-a curable diastolic heart failure. Nat Rev Cardiol. 2014; 11: 530-44.
- Nakanishi Y, Honda S, Yamano M, Kawasaki T, Yoshioka K. Constrictive pericarditis after SARS-CoV-2 vaccination: A case report. Int J
Infect Dis. 2022; 116: 238-240.
- Griessel R, Mitton B, Rule R, Said M. A case report of Nocardia
asiatica constrictive pericarditis in a patient with Human Immunodeficiency Virus. Cardiovasc Pathol. 2022; 58: 107403.
- George TJ, Arnaoutakis GJ, Beaty CA, Kilic A, Baumgartner WA, et
al. Contemporary etiologies, risk factors, and outcomes after pericardiectomy. Ann Thorac Surg. 2012; 94: 445-51.
- Bertog SC, Thambidorai SK, Parakh K, et al. Constrictive pericarditis: Etiology and cause-specific survival after pericardiectomy. J Am
Coll Cardiol. 2004; 43: 1445-52.
- Ling LH, Oh JK, Breen JF, et al. Calcific constrictive pericarditis: Is it
still with us? Ann Intern Med. 2000; 132: 444-50.
- Cho YH, Schaff HV, Dearani JA, et al. Completion pericardiectomy
for recurrent constrictive pericarditis: Importance of timing of recurrence on late clinical outcome of operation. Ann Thorac Surg.
2012; 93: 1236-40.