Introduction
Ranking fifth in morbidity and fourth in fatality in all malignancies, gastric cancer is one of the most prominent diseases world-wide [1]. Similarly, gastric cancer has made a great contribution
to the cancer burden in China. Gastric cancer is the second highest cancer to be diagnosed and the secondary consideration of
cancer-related death in China. As a transitioning country, China
bears a massive burden of the morbidity/mortality and five-year
prevalence rate for gastric cancer compared to most developed
countries [2]. Surgery-based multidisciplinary comprehensive
treatment remains the paramount approaches to selection for
treating gastric cancer [3]. An essential course of treatment for
non-metastatic gastric cancer is gastroplasty with lymph node dissection [4].
Gastrectomy offers a substantial risk of postoperative complications despite significant improvements in surgery and anesthetic procedures, postoperative care, and interventional radiology related to stomach cancer [5]. At the same time, recurrences
are common. In actuality, postoperative complications following
gastric surgery were reported to be 46% [6]. Undeniably, these
complications may reduce quality of life, postpone the start
of adjuvant treatment, and impede recovery [7]. Meanwhile, patients with conditions are at greater risk of disease recurrence [8].
Relevant evidence reveals that more than 70% of recurrence and
cancer-related mortality develop within two years of surgery, and
the recurrence and metastasis of gastric cancer can lead to a significant shortening of the time a patient survives [9].
Chronic and sustained inflammation of the gastric mucosa
has been demonstrated to not only act as a stimulant to the occurrence and advancement of gastric cancer [10], furthermore,
the inflammatory response stimulates and releases systemic cytokines, which attract the growth of remained cancer cells and
promote postoperative recurrence and metastasis [8]. A increasing variety of studies has revealed that several newly established
inflammation-based indicators: Neutrophil-To-Lymphocyte Ratio
(NLR), Lymphocyte-To-Monocytes Ratio (LMR), Lymphocyte-To-C
Reactive Protein Ratio (LCR) [11], including Fibrinogen-To-Albumin Ratio (FAR) [12] and SIRI [13] play an instrumental part in the
diagnosis, staging and prediction of gastric cancer. For example,
Fibrinogen-NLR (F-NLR) have served to prognostic prediction of
patients with esophageal-gastric junction and superior gastric
cancer after gastrectomy and have shown favourable predictive
effects [14]. Therefore, in order to further explore preoperative
indicators that can easily and accurately identify the risk of complication in the early post-operative period and recurrence or metastasis for patients undergoing radical gastrectomy, we propose
to combine SIRI and AFR these two biochemical markers with the
aim of improving the sensitivity of assessing inflammation, nutritional status and coagulation to improve the accuracy and specificity of predicting postoperative outcomes in short and long-term
for patients with gastric carcinoma.
Materials and methods
Patients and follow-up
We conducted retrospective research on patients at the Gansu
Provincial Hospital (Lanzhou, China) who had gastric cancer that
had been verified by histology from January 2018 to December 2019. The research protocols for the current investigation,
which conformed to the principles of the Declaration of Helsinki,
and received approval from the ethics board. Ethical consent:
21/10/2022-410, Gansu Provincial Hospital Medical Ethics Committee. From medical records, information was gathered on the
sex, age, tumor dimensions, tumor localization metastatic rate
of lymph nodes, degree of tumor differentiation, immunohisto-chemistry results (ki67, P53, Her2), TNM stage (refer to the American Joint Commission on Cancer (AJCC) gastric cancer TNM staging criteria (eighth edition)), ASA score, surgical approach, extent
of resection, duration of surgery, blood loss, periprocedural blood
transfusion, length of hospitalization, duration of postoperative
enteral nutrition. Inclusive criteria: (i) Between 18 and 80 years
of age with a clinically definite diagnosis of preoperative gastric
malignancy; (ii) Patients with postoperative pathological results
confirming primary gastric cancer; (iii) Patients undergoing D1/
D1+/D2 lymph node dissection with radical R0 resection for the
first time for radical gastric cancer. Exclusion criteria: (i) Patients
with distant tumor metastasis; (ii) Patients with combined hematological diseases, autoimmune diseases, infectious diseases or
liver dysfunction that may affect white blood cells; (iii) Preoperative neoadjuvant therapy (radiotherapy or chemotherapy); (iv)
Combined with other malignant tumors; (v) Incomplete data. Participants in the included studies were followed up by telephone
contact, outpatient review, hospitalization and others. Patients
were carefully followed-up every 3 to 6 months after surgery.
Annual follow-up was implemented after two years. Follow-up
outcomes were recurrence or metastasis within three years of
surgery. Postoperative recurrent metastases were defined as the
reappearance of malignancy associated with the primary lesion
or the presence of abdominal metastases, with or without distant
metastases, after radical resection. The last follow-up was processed in December 2022.
Laboratory variables and definition of fibrinogen albumin ratio and SIRI Index (SIRI-AFR)
Relevant indicators levels were assessed in blood samples
drawn within a week prior to the surgery. Retrospective analysis
and data collection from the electronic medical records included
additional parameters. In-hospital or within 30 days occurring
were categorized as early postoperative complications, and all
complications were graded for severity refer to the Clavien-Dindo
complication grading system [15], with Grade I or II complications
were categorized into minor complications, whereas Grade III and
higher were characterized as major complications. The general
post-operative pathology specimen’s lengthy diameter was used
to calculate the tumor size. Primary tumor locations were classified as upper, middle and lower stomach, accordingly. There were
two categories for differentiation level: badly differentiated and
moderately/well differentiated.
SIRI and AFR were calculated as the following: SIRI = Neutrophil count × Monocyte count/Lymphocyte count, AFR = Albumin/
Fibrinogen. Determined by the SIRI and AFR cut-off values, a scoring system was developed. Patients with a SIRI ≥ 1.007 and a AFR
≤9.849 were distributed a SIRI-AFR of 2, patients with a SIRI <1.007 and a AFR > 9.849 were allocated a SIRI-AFR score of 0, and
those with only one of SIRI ≥ 1.007 or AFR ≤ 9.849 were granted
a SIRI-AFR of 1.
Statistical analysis
All of the statistically analyzed were completed utilizing the
IBM SPSS for Windows, version 26.0 (IBM statistics for Windows,
version 26, IBM Corporation, Armonk, New York, United States).
Categorize material were indicated as n (number) and percentage (%), for normally distributed measures the information is described as the mean ± standard deviation and the non-normally
distributed continuous variables it is expressed as the median (Interquartile Range (IQR)). The paired groups were contrasted using
either the Mann Whitney-U test or the Student’s t-test, depending on the normality of the data distribution. The Chi-square test
was used to evaluate categorical group differences. To identify
factors affecting postoperative complications, logistic regression
models were employed. Receiver operating characteristic curves
with Youden indices were employed to establish the most favorable cut-off values for each outcome. Values of the Area Under
the Curve (AUC) were supplied with a 95% Confidence Interval
(CI). The Hazard Ratios (HRs) for disease recurrence or metastasis were calculated applying Cox proportional hazards models.
P<0.05 was designated as statistical significance.
Results
Patient characteristics
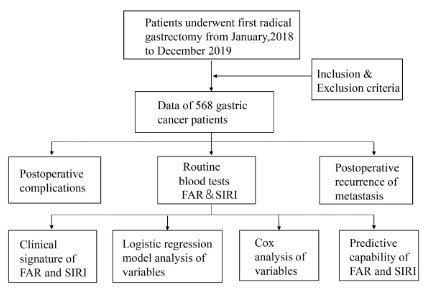
The flowchart for patient screening was displayed in Figure 1.
In total, 568 patients fit the inclusion criteria. No chemotherapy
or radiotherapy was administered to any of the patients prior to
surgery, and there was no perioperative mortality included. This
study included 442 men and 126 women. The average age of the
population group was 60.29 ± 9.79 (25-87). The average BMI ratio
prior surgery for all patients was 22.20 ± 3.37. 31.7% (n = 180)
of the patients were operated with open 40.0% (n = 227) with
laparoscopic approach and 28.3% (n = 161) with robot-assisted.
On the basis of the AJCC staging standards, 119 (21.0%) patients
were categorized as stage I, stage II patients accounted for 178
(31.3%), and stage III patients made up 271 (47.7%). No patient
was disregarded in the follow-up process. A median follow-up
time of 45 months was established for all patients, ranging from
12 to 61 months.
The clinical characterization of the study population was shown
in Table 1, along with a comparison of the characteristics and clinical aspects of the two group of patients who had no complications
(no) and/or experienced minor complications and those who had
major complications. The description of the features and clinicopathological comparison between the group of who did not experience recurrence or metastasis and the group of patients who did
recurrence or metastasis were displayed in Table 5.
Postoperative complications
A total of 89 (15.7%) patients in our statistics suffered serious
complications. The occurrence of early postoperative complications in individuals experiencing radical gastrectomy was showed
in Table 2. Complications included duration of enteral nutrition
was longer than 2 weeks in 26 patients, infection-related complications (incision infection, abdominal infection, pulmonary infection) in 234 patients, anastomotic fistula in 6 patients, pyloric or
intestinal obstruction in 14 patients, thrombosis or embolism in
15 patients, and seven patients developed postoperative shock,
they were all rescued after treatment.
In accordance with Table 1, age (p = 0.046), BMI (p = 0.003),
tumour size (<3/≥3 cm/) (p = 0.014), resection range (p = 0.019),
perioperative transfusion (p < 0.001), and hospital stay (p < 0.001)
were statistical significance between the two groups. For laboratory parameters, lymphocytes (p < 0.001), neutrophils (p < 0.001),
platelets (p = 0.013), monocytes (p = 0.032), albumin (p < 0.001),
fibrinogen (p < 0.001), CEA (p = 0.011), SIRI (p < 0.001) and AFR (p
< 0.001) also differed significantly between groups.
Correlations between SIRI, AFR and the clinicopathological
characteristics of gastric cancer
In accordance with the results in Table 3, Preoperative SIRI
level was related to the sex (p = 0.002), resection range (p = 0.008) among patients of gastric cancer. AFR had an association with the degree of tumor differentiation (p = 0.002)
and duration of enteral nutrition (p = 0.01). Meanwhile, both
preoperative conditions were related to age, tumour size (<3/≥3
cm), TNM stage, perioperative transfusion, CA199, CEA, amount
of bleeding, relapse or metastasis (p < 0.05).Upon further
analysis, among patients under 60 years of age, SIRI levels were
lower and AFR levels were higher (SIRI, p = 0.038; AFR, p < 0.001),
and SIRI levels were higher and AFR levels were lower in individu-
als with a maximum tumor diameter >3 cm (SIRI, p < 0.001; AFR, p
< 0.001). Furthermore, the level of SIRI in stage III was the highest
of the clinical stages, the level of AFR in stage III was the lowest
of the clinical stages (SIRI, p < 0.001; AFR, p < 0.001). For the peri-
operative blood transfusion patients, the level of SIRI was higher
and the level of AFR was lower (SIRI, p < 0.001; AFR, p < 0.001).
Group of CA199 and CEA positive patients, SIRI levels were higher
and AFR levels were lower (SIRI, p = 0.023, p < 0.001; AFR, p =
0.001 p < 0.001). The highest SIRI levels and lowest AFR levels
were observed in the group with intraoperative blood loss >400
ml (SIRI, p < 0.001; AFR, p < 0.001). The SIRI level of patients with
gastric cancer with relapse or metastasis was noticeably raised (p
< 0.001), and the AFR level was prominently reduced (p < 0.001).
Significance of preoperative SIRI and AFR levels for early serious postoperative complications in respectable gastric cancer
Table 4 listed the outcomes of the Univariate regression analysis that was executed to establish the OR values for the complication estimation. The result suggested that high preoperative SIRI
was substantially related with early serious postoperative complications (P < 0.001; HR 1.429; 95% CI 1.175-1.738). Meanwhile,
elevated preoperative AFR levels was a protective factor against
postoperative complications (P < 0.001; HR 0.729; 95% CI 0.665-0.799;). Additionally, age, BMI, tumor size (<3/≥3 cm), resection
range, perioperative transfusion and CEA (<5/≥5 ng/mL) were
other noteworthy variables revealed by univariate analysis (P <
0.05). Regards to multivariable analyses, preoperative SIRI and
AFR remained an independent influencing indicator for postoperative complications. (SIRI: P = 0.02; HR 1.222; 95% CI 1.031-1.446;
AFR: P < 0.001; HR 0.771; 95% CI 0.701-0.848). Furthermore, resection range (P=0.044; HR 1.682; 95% CI 1.015-2.787) and perioperative transfusion (P = 0.008; HR 2.028; 95% CI 1.202-3.422)
were other contributing factors.
Evaluation of predictive abilities for SIRI and AFR
Since the previous statistical findings concluded that high levels of AFR are a protective parameter for postoperative complications, in order to facilitate the calculation of the predictive power
of SIRI combined with AFR, we take fibrinogen to albumin ratio
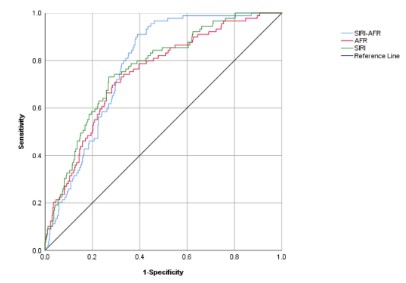
(the reciprocal of AFR) as the calculation amount. ROC curve generation and AUC calculation were used to determine the prediction capabilities of SIRI and AFR. The AUC values of SIRI, AFR, and
SIRI combine AFR levels were summarized in Figure 2. The AUC
values computed for the SIRI: AUC 0:765; 95% CI 0.714-0.815),
the AFR: AUC 0:743; 95%CI 0.689-0.797, the SIRI-AFR: AUC 0:779;
95% CI 0.737-0.820.
Establishment of the SIRI-AFR Score.
Based on the appropriate cut-off values for each determinant
which were established using receiver operating characteristic
curves with Youden’s index, patients were grouped accordingly.
Corresponding to the SIRI-AFR system, 219 (38.6%), 224 (39.4%),
and 125 (22.0%) patients had scores of 0, 1, and 2, accordingly.
The correlation between clinicopathological and relapse or
metastasis
The relationship between relapse or metastasis and clinicopathological factors was exhibited in Table 5. Recurrence or metastasis within 3 years in patients receiving radical resection of
gastric cancer was associated with these factors: Age (p = 0.019),
tumour location (p = 0.012), tumour size (<3/≥3 cm/) (p < 0.001),
differentiated degree (p = 0.033), Her-2 (p = 0.042), TNM stage (p
< 0.001), perioperative transfusion (p < 0.001), operation time (p
= 0.001) , lymph node metastasis rate (p < 0.001), lymphocytes (p
< 0.001), neutrophils (p < 0.001), platelets (p = 0.002), monocytes
(p < 0.001), albumin (p < 0.001), fibrinogen (p < 0.001), SIRI (p
< 0.001), AFR (p < 0.001), SIRI-AFR score (p < 0.001), CA199 (p
=0.001), CEA (p < 0.001), postoperative complication (p < 0.001)
and postoperative adjuvant chemotherapy (p = 0.012).
Univariate and multivariate Cox regression analysis for relapse or metastasis
Among patients of stomach carcinoma, univariate analyses
identified that the greater risk of relapse or metastasis was profoundly associated with correlated with older age (p = 0.009),
lower tumour location (p = 0.006), large tumor size (p < 0.001,
later clinical stage (p < 0.001), longer operating time (p = 0.002),
perioperative transfusion (p < 0.001), positive CA199 (p < 0.001),
positive CEA (p < 0.001), major postoperative complication (p <
0.001), no postoperative adjuvant chemotherapy was performed
(p = 0.006), high SIRI-AFR score (p < 0.001). Multivariate analysis revealed that TNM stage (p = 0.002; HR 5.100, 95% CI 1.847-
14.086), operation time (p = 0.029; HR 1.003, 95.0% CI 1.000-1.005), perioperative transfusion (p = 0.009; HR 1.660, 95.0% CI
1.135-2.428), positive CEA (p = 0.025; HR 1.528; 95% CI 1.054-2.213), postoperative adjuvant chemotherapy (p = 0.008; HR
0.475, 95% CI 0.273-0.826), SIRI-AFR score (p < 0.001; HR 4.363,
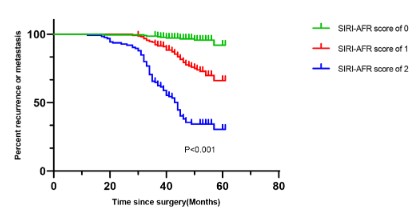
95% CI 2.170-9.037) were the independently determined prognostic variables for relapse or metastasis (Table 6). Further, as presented in Fig 3, we observed that the SIRI-AFR score could effectively differentiate patients into three distinguishing risk groups
for recurrence or metastasis.
Discussion
As a malignancy, gastric cancer seriously endangers public
health [16], and the occurrence of serious complications and recurrence and metastasis after surgery were still difficult problems
for clinicians. The development of gastric cancer was a multigene, multi-step process and certain key factors may participate
in the development of gastric cancer and even infiltration and metastasis at some stage. The systematic inflammatory response and
nutritional situation were two considerable contributing factors
[17]. SIRI and AFR were valuable novel procedures to evaluate the
inflammatory and nutritional condition of patients. To our knowledge, no studies have been done to examine how SIRI and AFR affect patients who received radical gastric cancer surgery in terms of early postoperative serious complications, early postoperative
recurrence or metastases. We created novel markers in the current study and evaluated their diagnostic and predictive potential
to aid in the early identification and therapy of gastric cancer.
Correa sequence, the canonical theory of cancer development in the stomach, indicated the inflammatory response was
an indispensable component in the tumor progression [17]. The
epidemiological and clinical investigations provided substantial
evidences that inflammation is associated with supporting the
growth of dissemination tumour cells [18]. Neutrophils, as an
essential element of tumor microenvironment, it participated in
tumor progression via multiple mechanisms, and activation of
neutrophils pathologically may symbolize the beginning of comprehension the procedures behind reactivation of dormant tumor
cells [19]. Moreover, neutrophils produced substances, such as
chemokines, cytokines, stromal degrading proteases and reactive
oxygen species, which can alter tumour growth and invasiveness.
Several studies have shown that neutrophils promote tumour
progression through stromal degradation and cancer cell proliferation [20]. Largely, neutrophil physiology at the cellular and molecular levels seems to determine that their primary function is
to facilitate transferential seeding. Neutrophil extracellular traps,
shaped by molecularly released DNA intended to capture tumor
cells in the circulation. Such an entanglement of circulating tumor
cells may be beneficial to intraluminal survival, adhesion to endothelium, and extravasation [21].
Table 1: Patient baseline characteristics and their correlations with complications after resectable gastric cancer.
| Variables |
Minor/no complication n = 479 (84.3%) |
Major complication n = 89 (15.7%) |
P values |
| Gender |
Male |
378(78.9%) |
64(71.9%) |
0.144a |
|
Female |
101(21.1%) |
25(28.1%) |
|
| Age (y) |
|
59.93±9.66 |
62.19±10.33 |
0.046b |
| Underlying disease |
No |
382(79.7%) |
75(84.3%) |
0.323a |
|
Yes |
97(20.3%) |
14(15.7%) |
|
| BMI |
|
22.37±3.36 |
21.23±3.28 |
0.003b |
| Tumour location |
Upper third |
56(11.7%) |
15(16.9%) |
0.537a |
|
Middle third |
44(9.2%) |
8(9.0%) |
|
|
Lower third |
377(78.7%) |
66(74.2%) |
|
| Tumor dimensions (cm) |
<3 |
148(30.9%) |
16(18.0%) |
0.014a |
|
≥3 |
331(69.1%) |
73(82.0%) |
|
| Differentiation |
Moderate and poor |
460(96.0%) |
86(96.6%) |
1a |
|
Well |
19(4.0%) |
3(3.4%) |
|
| TNM stage |
I |
106(22.1%) |
13(14.6%) |
0.236a |
|
II |
150(31.3%) |
28(31.5%) |
|
|
III |
223(46.6%) |
48(53.9%) |
|
| Approach |
Open |
149(31.1%) |
31(34.8%) |
0.539a |
|
Laparoscopic |
190(39.7%) |
37(41.6%) |
|
|
Robot-assisted |
140(29.2%) |
21(23.6%) |
|
| Operation |
Subtotal gastrectomy |
248(51.8%) |
34(38.2%) |
0.019a |
|
Total gastrectomy |
231(48.2%) |
55(61.8%) |
|
| ASA |
I-II |
433(90.4%) |
76(85.4%) |
0.155a |
|
III-IV |
46(9.6%) |
13(14.6%) |
|
| Blood loss (ml) |
|
100(100) |
150(200) |
0.089c |
| Duration of surgery (minutes) |
|
240(90) |
250(85) |
0.079a |
| Perioperative transfusion |
No |
389(81.2%) |
50(56.2%) |
<0.001a |
| |
Yes |
90(18.8%) |
39(43.8%) |
|
| Length of hospitalization (days) |
|
17.00(5.00) |
21.00(8.00) |
<0.001c |
| Lymph node metastasis rate (%) |
|
4.02%±15.17% |
4.52%±14.72% |
0.264b |
| Lymphocytes (×109/L) |
|
1.44(0.72) |
1.18(0.59) |
<0.001c |
| Neutrophils (×109/L) |
|
3.52(1.76) |
5.03(1.56) |
<0.001c |
| Platelet (×109/L) |
|
213(86) |
234(107) |
0.013c |
| Monocyte (×109/L) |
|
0.39(0.16) |
0.42(0.17) |
0.032c |
| Albumin (g/L) |
|
39.40±4.36 |
36.60±4.50 |
<0.001b |
| Fibrinogen (g/L) |
|
3.39±0.79 |
4.08±1.04 |
<0.001b |
| SIRI |
|
0.95(0.82) |
1.54(0.97) |
<0.001c |
| AFR |
|
12.32±3.46 |
9.54±2.68 |
<0.001c |
| CA199 (ng/mL) |
|
406(84.8%) |
69(77.5%) |
0.090a |
| |
|
73(15.2%) |
20(22.5%) |
|
| CEA (ng/mL) |
|
386(80.6%) |
61(68.5%) |
0.011a |
| |
|
93(19.4%) |
28(31.5%) |
|
The bold numbers in the tables are P-values with statistical significance (<0.05).
aChi-square test, bStudent’s t-test with mean ± standard deviation, cMann-Whitney U test with median (interquartile range), SD: Standard deviation,
IQR: Interquartile range, BMI: Body mass index, ASA: American society of anesthesiology. SIRI: Systemic Inflammation Response Index; AFR: Albumin
Fibrinogen Ratio; CA199: Carbohydrate Antigen 199; CEA: Carcinoembryonic Antigen.
Table 2: Occurrence of short-term postoperative complications in
patients undergoing radical gastric cancer resection.
| Postoperative complications |
N (%) |
| Enteral nutritiontime>2 weeks |
26(4.58%) |
| Incision infection |
4(0.70%) |
| Abdominal infection |
160(28.17%) |
| Pulmonary infection |
70(12.32%) |
| Pelvic effusion |
6(1.06%) |
| Abdominal bleeding |
9(1.58%) |
| Anastomotic fistula |
6(1.06%) |
| Pyloric or intestinal obstruction |
14(2.46%) |
| Deepvenous thrombosis |
10(1.76%) |
| Splenic embolism |
1(0.18%) |
| Pulmonary embolism |
4(0.70%) |
| Shock |
7(1.23%) |
Table 3: Clinicopathological variables and preoperative SIRI and AFR in gastric cancer patients.
| Variables |
|
Preoperative SIRI (Median(IQR) |
*P values |
Preoperative AFR(mean±SD) |
**P values |
| Gender |
Male |
1.11(0.89) |
0.002 |
11.96±3.68 |
0.252 |
| |
Female |
0.91(0.87) |
|
11.62±2.77 |
|
| Age (y) |
<60 |
1.03(0.86) |
0.038 |
12.58±3.83 |
<0.001 |
| |
≥60 |
1.14(0.96) |
|
11.26±3.04 |
|
| Underlying disease |
No |
1.07(0.90) |
0.187 |
11.88±3.55 |
0.989 |
| |
Yes |
1.14(0.91) |
|
11.89±3.31 |
|
| BMI |
<24 |
1.06(0.89) |
0.29 |
11.82±3.60 |
0.464 |
| |
≥24 |
1.15(0.85) |
|
12.06±3.23 |
|
| Tumour location |
Upper third |
1.17(1.09) |
0.164 |
11.63±3.09 |
0.695 |
| |
Middle third |
1.00(0.80) |
|
12.36±3.14 |
|
| |
Lower third |
1.07(0.88) |
|
11.88±3.61 |
|
| Tumor dimensions (cm) |
<3 |
0.89(0.69) |
<0.001 |
13.44±3.86 |
<0.001 |
| |
≥3 |
1.17(0.92) |
|
11.26±3.13 |
|
| Differentiation |
Moderate and poor |
1.08(0.90) |
0.235 |
11.80±3.46 |
0.002 |
| |
Well |
0.91(0.71) |
|
14.11±3.90 |
|
| TNM stage |
I |
0.78(0.58) |
<0.001 |
13.70±4.20 |
<0.001 |
| |
II |
1.08(0.93) |
|
11.66±3.41 |
|
| |
III |
1.20(0.92) |
|
11.24±2.90 |
|
| Approach |
Open |
1.16(0.89) |
0.261 |
12.05±3.97 |
0.617 |
| |
Laparoscopic |
1.05(0.87) |
|
11.90±3.29 |
|
| |
Robot-assisted |
1.08(0.97) |
|
11.68±3.22 |
|
| Operation |
Subtotal gastrectomy |
0.97(0.84) |
0.008 |
12.10±3.41 |
0.169 |
| |
Total gastrectomy |
1.15(0.94) |
|
11.67±3.58 |
|
| ASA |
I-II |
1.07(0.90) |
0.458 |
11.89±3.53 |
0.906 |
| |
III-IV |
1.23(0.85) |
|
11.83±3.24 |
|
| Perioperative transfusion |
No |
1.02(0.81) |
<0.001 |
12.37±3.50 |
<0.001 |
| |
Yes |
1.44(1.20) |
|
10.25±3.00 |
|
| CA199 (ng/mL) |
Negative |
1.06(0.86) |
0.023 |
12.14±3.16 |
0.001 |
| |
Postive |
1.23(1.17) |
|
10.57±2.83 |
|
| CEA (ng/mL) |
Negative |
1.03(0.83) |
<0.001 |
12.24±3.59 |
<0.001 |
| |
Postive |
1.29(1.30) |
|
10.58±2.81 |
|
| Blood loss (ml) |
<200 |
1.02(0.77) |
0.011 |
12.24±3.63 |
0.013 |
| |
200≤X≤400 |
1.16(1.02) |
|
11.49±3.32 |
|
| |
>400 |
1.25(0.95) |
|
11.00±3.00 |
|
| Relapse or metastasis |
No |
0.93(0.82) |
<0.001 |
12.65±3.35 |
<0.001 |
| |
Yes |
1.48(1.07) |
|
9.40±2.77 |
|
| P53 |
Wild |
1.14(0.94) |
0.372 |
11.89±3.40 |
0.997 |
| |
Mutant |
1.06(0.82) |
|
11.89±3.56 |
|
| Ki-67 |
0%-49% |
0.92(0.79) |
0.183 |
12.58±3.16 |
0.249 |
| |
50%-74% |
1.14(0.79) |
|
11.87±3.30 |
|
| |
75%-100% |
1.07(0.95) |
|
11.76±3.66 |
|
| Her-2 |
Negative |
1.08(0.91) |
0.795 |
11.88±3.53 |
0.891 |
| |
Postive |
0.98(0.75) |
|
11.95±3.19 |
|
| Lymph node metastasis rate(%) |
<4.60% |
1.07(0.90) |
0.471 |
11.95±3.55 |
0.112 |
| |
≥4.60% |
1.20(0.83) |
|
11.10±2.81 |
|
| Enteral nutrition time |
≤7 days |
1.06(0.91) |
0.087 |
12.18±3.62 |
0.01 |
| |
>days |
1.15(0.88) |
|
11.40±3.23 |
|
The bold numbers in the tables are P-values with statistical significance (<0.05).
*p-value using Mann-Whitney U test with median (IQR), **p-value using Student’s t-test with mean ± standard deviation. SD: Standard devia-
tion, IQR: Interquartile range, BMI: Body mass index, ASA: American society of anesthesiology, CA199: Carbohydrate antigen 199, CEA: carcino-
embryonic antigen SIRI: Systemic inflammation response index; AFR: Albumin fibrinogen ratio.
Table 4: Univariate and multivariate analyses of the logistic regression model for postoperative complications in patients.
| Variables |
Univariate analysis |
Multivariate analysis |
| OR |
95% CI |
P |
OR |
95% CI |
P |
| Age (y) |
1.025 |
(1.000-1.049) |
0.046 |
1.003 |
(0.977-1.030) |
0.819 |
| BMI |
0.896 |
(0.832-0.964) |
0.003 |
0.928 |
(0.857-1.005) |
0.068 |
| Tumor dimensions (cm) |
2.04 |
(1.148-3.624) |
0.015 |
1.003 |
(0.523-1.926) |
0.992 |
| Operation |
1.737 |
(1.092-2.761) |
0.02 |
1.682 |
(1.015-2.787) |
0.044 |
| Perioperative transfusion |
3.371 |
(2.091-5.434) |
<0.001 |
2.028 |
(1.202-3.422) |
0.008 |
| CEA (ng/mL) |
1.905 |
(1.154-3.146) |
0.012 |
1.213 |
(0.696-2.112) |
0.496 |
| SIRI |
1.429 |
(1.175-1.738) |
<0.001 |
1.222 |
(1.031-1.446) |
0.02 |
| AFR |
0.729 |
(0.665-0.799) |
<0.001 |
0.771 |
(0.701-0.848) |
<0.001 |
The bold numbers in the tables are P-values with statistical significance (<0.05).
BMI: Body mass index, SIRI: Systemic inflammation response index; AFR: Albumin fibrinogen ratio.
Table 5: Patient baseline characteristics and their correlations with relapse or metastasis in 3 years after surgery.
| Variables |
No relapse or metastasis n = 435 (76.6%) |
Relapse or metastasis n = 133 (23.4%) |
P values |
| Gender |
Male |
333(76.6%) |
109(82.0%) |
0.189a |
| |
Female |
102(23.4%) |
24(18.0%) |
|
| Age (y) |
|
59.75±9.51 |
62.02±10.51 |
0.019b |
| Underlying disease |
No |
350(80.5%) |
107(80.5%) |
0.998a |
| |
Yes |
85(19.5%) |
26(19.5%) |
|
| BMI |
|
22.24±3.39 |
22.03±3.30 |
0.524b |
| Tumour location |
Upper third |
48(11.0%) |
23(17.3%) |
0.012a |
| |
Middle third |
48(11.0%) |
4(3.0%) |
|
| |
Lower third |
337(77.5%) |
106(79.7%) |
|
| Tumor dimensions (cm) |
<3 |
145(33.3%) |
19(14.3%) |
<0.001a |
| |
≥3 |
290(66.7%) |
114(85.7%) |
|
| Differentiation |
Moderate and poor |
414(95.2%) |
132(99.2%) |
0.033a |
| |
Well |
21(4.8%) |
1(0.8%) |
|
| P53 |
Wild |
157(36.1%) |
48(36.1%) |
1a |
| |
Mutant |
278(63.9%) |
85(63.9%) |
|
| Ki-67 |
0%-49% |
49(11.3%) |
11(8.3%) |
0.513a |
| |
50%-74% |
134(30.8%) |
46(34.6%) |
|
| |
75%-100% |
140(29.2%) |
21(23.6%) |
|
| Her-2 |
Negative |
395(90.8%) |
128(96.2%) |
0.042a |
| |
Postive |
40(9.2%) |
5(3.8%) |
|
| TNM stage |
I |
112(25.7%) |
7(5.3%) |
<0.001a |
| |
II |
145(33.3%) |
33(24.8%) |
|
| |
III |
178(40.9%) |
93(69.9%) |
|
| Approach |
Open |
135(31.0%) |
45(33.8%) |
0.099a |
| |
Laparoscopic |
190(39.7%) |
37(41.6%) |
|
| |
Robot-assisted |
116(26.7%) |
45(33.8%) |
|
| Operation |
Subtotal gastrectomy |
223(51.3%) |
59(44.4%) |
0.163a |
| |
Total gastrectomy |
212(48.7%) |
74(55.6%) |
|
| ASA |
I-II |
394(90.6%) |
115(86.5%) |
0.174a |
| |
III-IV |
41(9.4%) |
18(13.5%) |
|
| Blood loss (ml) |
<200 |
258(59.3%) |
72(54.1%) |
0.485a |
| |
200≤X≤400 |
145(33.3%) |
48(36.1%) |
|
| |
>400 |
32(7.4%) |
13(59.8%) |
|
| Perioperative transfusion |
No |
359(82.5%) |
80(60.2%) |
<0.001a |
| |
Yes |
76(17.5%) |
53(39.8%) |
|
| Enteral nutrition time (days) |
|
7.00(4.00) |
7.00(3.00) |
0.149a |
| Duration of surgery (minutes) |
|
240(80) |
260(95) |
0.001c |
| Length of hospitalization (days) |
|
17.00(6.00) |
18.00(7.00) |
0.157c |
| Lymph node metastasisrate (%) |
|
0.06%(0.33%) |
0.27%(0.70%) |
<0.001c |
| Lymphocytes (×109/L) |
|
1.44(0.71) |
1.20(0.56) |
<0.001c |
| Neutrophils (×109/L) |
|
3.52(1.90) |
4.32(1.54) |
<0.001c |
| Platelet (×109/L) |
|
209(86) |
228(93) |
0.002c |
| Monocyte (×109/L) |
|
0.38(0.17) |
0.44(0.17) |
<0.001c |
| Albumin (g/L) |
|
39.68±3.99 |
36.59±6.18 |
<0.001b |
| Fibrinogen (g/L) |
|
3.30±0.71 |
4.14±1.01 |
<0.001b |
| SIRI |
|
0.93(0.82) |
1.48(1.07) |
<0.001c |
| AFR |
|
12.65±3.35 |
9.40±2.76 |
<0.001b |
| SIRI-ARF score |
0 |
210(48.3%) |
9(6.8%) |
<0.001a |
| |
1 |
175(40.2%) |
49(36.8%) |
|
| |
2 |
50(11.5%) |
75(56.4%) |
|
| CA199 (ng/mL) |
Negative |
376(86.4%) |
99(74.4%) |
0.001a |
| |
Postive |
59(13.6%) |
34(25.6%) |
|
| CEA (ng/mL) |
Negative |
361(83.0%) |
86(64.7%) |
<0.001a |
| |
Postive |
74(17.0%) |
47(35.3%) |
|
| Postoperative complication |
Minor/no |
390(89.7%) |
89(66.9%) |
<0.001a |
| |
Major |
45(10.3%) |
44(33.1%) |
|
| Postoperative chemotherapy |
No |
130(29.9%) |
25(18.8%) |
0.012a |
| |
Yes |
305(70.1%) |
108(81.2%) |
|
The bold numbers in the tables are P-values with statistical significance (<0.05).
aChi-square test, bStudent’s t-test with mean ± standard deviation, cMann-Whitney U test with median (interquartile range), SD: Standard
deviation, IQR: Interquartile range, BMI: Body mass index, ASA: American society of anesthesiology. SIRI: Systemic inflammation response
index; AFR: Albumin fibrinogen ratio, CA199: Carbohydrate antigen 199, CEA: Carcinoembryonic antigen.
Table 6: Univariate and multivariate Cox regression analyses for relapse or metastasis in patients with gastric cancer.
| Variables |
Univariate analysis |
|
Multivariate analysis |
|
|
OR |
95% CI |
P |
OR |
95% CI |
P |
| Age (y) |
|
1.025 |
(1.006-1.044) |
0.009 |
1.008 |
(0.990-1.027) |
0.381 |
| Tumour location |
|
|
|
0.053 |
|
|
0.231 |
|
Upper 1/3 |
0.224 |
(0.077-0.647) |
0.006 |
0.451 |
(0.153-1.336) |
0.151 |
|
Middle 1/3 |
0.843 |
(0.537-1.323) |
0.391 |
1.239 |
(0.774-1.985) |
0.372 |
|
Low 1/3 |
Ref |
|
|
Ref |
|
|
| Tumor dimensions (cm) |
|
|
|
|
|
|
|
|
<3/≥3 |
3.002 |
(1.845-4.884) |
<0.001 |
1.428 |
(0.835-2.444) |
0.193 |
| Differentiation |
|
|
|
|
|
|
|
|
Moderate and poor/ Well |
0.159 |
(0.022-1.134) |
0.067 |
0.667 |
(0.081-5.491) |
0.707 |
| Her-2 |
|
|
|
|
|
|
|
|
Negative/Positive |
2.151 |
(0.880-5.258) |
0.093 |
0.520 |
(0.209-1.295) |
0.160 |
| TNM stage |
|
|
|
|
|
|
<0.001 |
|
I |
Ref |
|
|
Ref |
|
|
|
II |
3.533 |
(1.563-7.989) |
0.002 |
1.704 |
(0.634-4.576) |
0.291 |
|
III |
7.427 |
(3.443-16.022) |
<0.001 |
5.100 |
(1.847-14.086) |
0.002 |
| Operation time (minutes) |
|
1.004 |
(1.001-1.006) |
0.002 |
1.003 |
(1.000-1.005) |
0.029 |
| Perioperative transfusion |
|
|
|
|
|
|
|
|
No/Yes |
2.65 |
(1.872-3.752) |
<0.001 |
1.660 |
(1.135-2.428) |
0.009 |
| CA199 (ng/mL) |
|
|
|
|
|
|
|
|
Negative/Positive |
2.039 |
(1.380-3.013) |
<0.001 |
1.417 |
(0.942-2.130) |
0.094 |
| CEA (ng/mL) |
|
|
|
|
|
|
|
|
Negative/Positive |
2.198 |
(1.540-3.137) |
<0.001 |
1.528 |
(1.054-2.213) |
0.025 |
| Lymph node metastasis rate (%) |
|
1.001 |
(0.991-1.012) |
0.790 |
0.997 |
(0.986-1.009) |
0.666 |
| Postoperative complication |
|
|
|
|
|
|
|
|
No or Minor/Major |
3.35 |
(2.331-4.813) |
<0.001 |
1.220 |
(0.820-1.815) |
0.327 |
| Postoperative chemotherapy |
|
|
|
|
|
|
|
|
No/Yes |
1.850 |
(1.197-2.859) |
0.006 |
0.475 |
(0.273-0.826) |
0.008 |
| SIRI-AFR score |
|
|
|
|
|
|
|
|
0 |
Ref |
|
|
Ref |
|
|
|
1 |
6.057 |
(2.975-12.334) |
<0.001 |
4.363 |
(2.107-9.037) |
<0.001 |
|
2 |
22.705 |
(11.354-45.402) |
<0.001 |
12.554 |
(5.995-26.291) |
<0.001 |
The bold numbers in the tables are P-values with statistical significance (<0.05).
CA199: Carbohydrate Antigen 199; CEA: Carcinoembryonic Antigen; SIRI: Systemic Inflammation Response Index; AFR: Albumin Fibrinogen Ratio.
Monocytes serve as cells bridging the innate and adaptive immunity, they can promote cancer immune escape by differentiation into immunomodulatory cells [22]. Factually, certain mutual
interactions between circulation of carcinoma cells and circulating monocytes can effectively accelerate their dissemination and
extravasation at distant sites [23]. They can have an immediate
involvement in promotion, support and maintenance of tumour
growth by affecting the tumor microenvironment through multiple mechanisms that produce tolerance, angiogenesis and accelerated tumor cell proliferation [24].
Lymphocytes played a part in immunologic surveillance and
were contributory to identification and destruction [25]. Importantly, a biochemical alteration of T cells can modulate cellular
activities and promote tumor progression [26]. With evidence
that the magnitude and composition of tumour infiltrating lymphocytes can affect survival of oesophageal adenocarcinoma [27].
The abnormal fibrinogen levels can lead to disturbances in
the control of normal homeostasis during coagulation. And quite
possibly, sedimentation of fibrinogen on cancer cells can form a
physical shield to protect cancer cells from recognition and lysis
by NK cells [28].
The level of albumin is influenced by nutritional status and
metabolism. Hypoalbuminemia can generate immunodeficiency
in tumour patients, which reduces the effectiveness of treatment
and increases mortality [29]. As such, albumin levels were a recognized prognostic factor for a number of malignancies [30,31].
Similarly, some research suggested that albumin levels affect the
likelihood of postoperative complications and cancer recurrence
[32,33].
Furthermore, mounting data pointed to the usefulness of
SIRI as a predictor of adverse survival in patients with a range of
malignancies, including gastric cancer [34-36]. According to our
findings, SIRI constituted an independently attributable risk for
severe postoperative complications in patients with radical gastrectomy. Recently, Mario and his colleagues confirmed that SIRI
can be considered to potentially predict anastomotic fistula after
total gastrectomy [37]. Similarly, related research has also demonstrated that AFR can predict patients with pancreatic cancer [30],
gallbladder cancer [38], and colorectal cancer [39] prognosis. Our
findings suggested that AFR was also a worthwhile parameter
for predicting serious complications and recurrent metastases
in patients receiving radical gastrectomy in the early postoperative period. The predictive value of combining SIRI and AFR for
early postoperative serious complications and recurrent metastases in patients undergoing radical gastrectomy was first identified through our study, and it was an encouraging tool for cancer
treatment strategy decisions.
In particular, the surgical resection range was also discovered
in our study to be a risk factor for early complications following radical gastric cancer resection. Total gastrectomy significantly
damaged the digestive system and had systemic repercussions,
which warned us of the importance and necessity of early discovery, diagnosis and treatment of gastric cancer. Interestingly, we
observed perioperative blood transfusion to be a contributing factor for early recurrence metastases as well as postoperative problems. A growing body of research suggested that transfusions of
allogeneic blood may have immunomodulatory impact that lowered the threshold for periprosthetic infections through a number
of mechanisms, including decreased natural killer cell activity, an
imbalance in the normal distribution of helper and/or suppressor
T cells and improper antigen presentation by host cells [40]. In
fact, there were also studies have shown that perioperative blood
transfusion can increase the chance of postoperative infection
[41], and was associated with complications after gastrectomy
[42]. Our results were consistent with those observed by Stephen
T McSorley [43] and Xiaowen Liu [44], who noted perioperative
blood transfusion is linked to worse survival following surgery for
colorectal cancer and gastric cancer. Furthermore, postoperative
adjuvant chemotherapy was also a noteworthy factor affecting
recurrent metastasis. However, when assessing the risks and advantages of treatment, adverse effects of chemotherapy may be a
crucial consideration. During our follow-up, we learned that many
patients did not complete the regular chemotherapy cycle due to
adverse reactions such as nausea and vomiting after chemotherapy, which is a question worth pondering.
A few limitations applied to this investigation. Firstly, the retrospective nature of the study at a single institution restricts its
statistical power. Subsequently, we lacked evaluation of postoperative SIRI and AFR dynamic changes in a relatively large cohort
of GC patients, larger multicenter prospective randomized controlled trials are needed to verify our conclusion. Finally, despite
the fact that SIRI and AFR are worthwhile and easily obtainable
routine blood parameters, the underlying biological and molecular mechanisms that account for their prognostic and predictive
nature remain unclear.
Conclusion
Overall, the findings of this investigation indicate a significant
association between preoperative SIRI and AFR in individuals with
gastric cancer and the occurrence of severe complications, as well
as early postoperative recurrence or metastasis. These results
may aid surgeons and oncologists in conducting more effective
preoperative evaluations and management, and developing post-operative monitoring plans for patients with gastric cancer.
Abbreviations
SIRI: Systemic Inflammatory Response Index; AFR: Albumin
Fibrinogen Ratio; NLR: Neutrophil-To-Lymphocyte Ratio; LMR:
Lymphocyte-To-Monocytes Ratio; LCR: Lymphocyte-To-C Reactive Protein Ratio; FAR: Fibrinogen-To-Albumin Ratio; F-NLR: Fibrinogen- Neutrophil-To-Lymphocyte Ratio; AJCC: American Joint
Commission On Cancer; IQR: Interquartile Range; CA199: Carbohydrate Antigen 199; CEA: Carcinoembryonic Antigen; AUC: Area
Under The Curve; CI: Confidence Interval; HR: Hazard Ratio.
Declarations
Ethical approval: The research protocols for the current investigation, which conformed to the principles of the Declaration of
Helsinki, and received approval from the ethics board. Ethical consent: 21/10/2022-410, Gansu Provincial Hospital Medical Ethics
Committee.
Data availability statement: The datasets used and/or analyzed
during the current study are available from the corresponding author on reasonable request.
Conflict of interest: The authors declared no conflict of interest
in the publication of this paper.
Contributors: JR conceived and designed the study and revised
the manuscript. JR, DW, LZ, SL, and MY conducted all data collection and analysis and compiled charts. All authors read and approved the final manuscript.
References
- Sung H, Ferlay J, Siegel RL, et al. Global Cancer Statistics 2020:
GLOBOCAN Estimates of Incidence and Mortality Worldwide for
36 Cancers in 185 Countries. CA Cancer J Clin. 2021; 71: 209-49.
- Cao W, Chen HD, Yu YW, et al. Changing profiles of cancer burden
worldwide and in China: A secondary analysis of the global cancer
statistics 2020. Chin Med J (Engl). 2021; 134: 783-91.
- Joshi SS, Badgwell BD. Current treatment and recent progress in
gastric cancer. CA Cancer J Clin. 2021; 71: 264-79.
- Cravo M, Fidalgo C, Garrido R, et al. Towards curative therapy in
gastric cancer: Faraway, so close! World J Gastroenterol. 2015; 21:
11609-20.
- Kanda M, Ito S, Mochizuki Y, et al. Multi-institutional analysis of
the prognostic significance of postoperative complications after
curative resection for gastric cancer. Cancer Med. 2019; 8:5 194-201.
- Yildirim M, Koca B. Lymphocyte C-reactive protein ratio: A new
biomarker to predict early complications after gastrointestinal oncologic surgery. Cancer Biomark. 2021; 31: 409-17.
- Kanda M. Preoperative predictors of postoperative complications
after gastric cancer resection. Surg Today. 2020; 50: 3-11.
- Eto K, Hiki N, Kumagai K, et al. Prophylactic effect of neoadjuvant
chemotherapy in gastric cancer patients with postoperative complications. Gastric Cancer. 2018; 21: 703-09.
- Kang WM, Meng QB, Yu JC, et al. Factors associated with early recurrence after curative surgery for gastric cancer. World J Gastroenterol. 2015; 21: 5934-40.
- Zavros Y, Merchant JL. The immune microenvironment in gastric
adenocarcinoma. Nat Rev Gastroenterol Hepatol. 2022; 19: 451-67.
- Caglar R. The relationship of different preoperative inflammatory
markers with the prognosis of gastric carcinoma. Asian J Surg.
2022.
- Lin GT, Chen QY, Zhong Q, et al. ASO Author Reflections: Fibrinogen-Albumin Ratio as New Promising Biochemical Marker for Predicting Oncological Outcomes in Gastric Cancer Compared with
the Combination of Other Inflammation-Related Factors. Ann Surg Oncol. 2021; 28: 7074-75.
- Liu Z, Ge H, Miao Z, et al. Dynamic Changes in the Systemic Inflammation Response Index Predict the Outcome of Resectable Gastric
Cancer Patients. Front Oncol. 2021; 11: 577043.
- Cong X, Li S, Zhang Y, et al. The combination of preoperative fibrinogen and neutrophil-lymphocyte ratio is a predictive prognostic factor in esophagogastric junction and upper gastric cancer. J
Cancer. 2019; 10: 5518-26.
- Kim W, Kim HH, Han SU, et al. Decreased Morbidity of Laparoscopic Distal Gastrectomy Compared With Open Distal Gastrectomy for
Stage I Gastric Cancer: Short-term Outcomes From a Multicenter
Randomized Controlled Trial (KLASS-01). Ann Surg. 2016; 263: 28-35.
- Mihmanli M, Ilhan E, Idiz UO, et al. Recent developments and innovations in gastric cancer. World J Gastroenterol. 2016; 22: 4307-20.
- Banks M, Graham D, Jansen M, et al. British Society of Gastroenterology guidelines on the diagnosis and management of patients
at risk of gastric adenocarcinoma. Gut. 2019; 68: 1545-75.
- Qian BZ. Inflammation fires up cancer metastasis. Semin Cancer
Biol. 2017; 47: 170-76.
- Perego M, Tyurin VA, Tyurina YY, et al. Reactivation of dormant
tumor cells by modified lipids derived from stress-activated neutrophils. Sci Transl Med. 2020; 12.
- Galdiero MR, Marone G, Mantovani A. Cancer Inflammation and
Cytokines. Cold Spring Harb Perspect Biol. 2018; 10.
- Cools-Lartigue J, Spicer J, McDonald B, et al. Neutrophil extracellular traps sequester circulating tumor cells and promote metastasis.
J Clin Invest 2013; 123: 3446-58.
- Gabrilovich DI, Ostrand-Rosenberg S, Bronte V. Coordinated regulation of myeloid cells by tumours. Nat Rev Immunol. 2012; 12:
253-68.
- Lambert AW, Pattabiraman DR, Weinberg RA. Emerging Biological
Principles of Metastasis. Cell 2017; 168: 670-91.
- Kim IS, Gao Y, Welte T, et al. Immuno-subtyping of breast cancer
reveals distinct myeloid cell profiles and immunotherapy resistance mechanisms. Nat Cell Biol. 2019; 21: 1113-26.
- Farag CM, Antar R, Akosman S, et al. What is hemoglobin, albumin,
lymphocyte, platelet (HALP) score? A comprehensive literature review of HALP’s prognostic ability in different cancer types. Oncotarget. 2023; 14: 153-72.
- Jiang S, Yan W. T-cell immunometabolism against cancer. Cancer
Lett. 2016; 382: 255-58.
- Noble F, Mellows T, McCormick Matthews LH, et al. Tumour infiltrating lymphocytes correlate with improved survival in patients
with oesophageal adenocarcinoma. Cancer Immunol Immunother.
2016; 65: 651-62.
- Palumbo JS, Talmage KE, Massari JV, et al. Platelets and fibrin(ogen)
increase metastatic potential by impeding natural killer cell-mediated elimination of tumor cells. Blood. 2005; 105: 178-85.
- Cham S, Chen L, St Clair CM, et al. Development and validation
of a risk-calculator for adverse perioperative outcomes for women
with ovarian cancer. Am J Obstet Gynecol. 2019; 220: 571.e1-71.
e8.
- Fang L, Yan FH, Liu C, et al. Systemic Inflammatory Biomarkers, Especially Fibrinogen to Albumin Ratio, Predict Prognosis in Patients
with Pancreatic Cancer. Cancer Res Treat. 2021; 53: 131-39.
- Liao CK, Yu YL, Lin YC, et al. Prognostic value of the C-reactive protein to albumin ratio in colorectal cancer: an updated systematic
review and meta-analysis. World J Surg Oncol. 2021; 19: 139.
- Bullock AF, Greenley SL, McKenzie GAG, et al. Relationship between markers of malnutrition and clinical outcomes in older
adults with cancer: systematic review, narrative synthesis and
meta-analysis. Eur J Clin Nutr. 2020; 74: 1519-35.
- Maumy L, Harrissart G, Dewaele P, et al. [Impact of nutrition on
breast cancer mortality and risk of recurrence, a review of the evidence]. Bull Cancer. 2020; 107: 61-71.
- Qi Q, Zhuang L, Shen Y, et al. A novel systemic inflammation response index (SIRI) for predicting the survival of patients with pancreatic cancer after chemotherapy. Cancer. 2016; 122: 2158-67.
- Sun L, Hu W, Liu M, et al. High Systemic Inflammation Response
Index (SIRI) Indicates Poor Outcome in Gallbladder Cancer Patients
with Surgical Resection: A Single Institution Experience in China.
Cancer Res Treat. 2020; 52: 1199-210.
- Xu L, Yu S, Zhuang L, et al. Systemic inflammation response index
(SIRI) predicts prognosis in hepatocellular carcinoma patients. Oncotarget 2017; 8: 34954-60.
- Schietroma M, Romano L, Schiavi D, et al. Systemic inflammation
response index (SIRI) as predictor of anastomotic leakage after total gastrectomy for gastric cancer. Surg Oncol. 2022; 43: 101791.
- Xu WY, Zhang HH, Xiong JP, et al. Prognostic significance of the
fibrinogen-to-albumin ratio in gallbladder cancer patients. World
J Gastroenterol. 2018; 24: 3281-92.
- Sun F, Tan YA, Gao QF, et al. Circulating fibrinogen to pre-albumin
ratio is a promising biomarker for diagnosis of colorectal cancer. J
Clin Lab Anal. 2019; 33: e22635.
- Raghavan M, Marik PE. Anemia, allogenic blood transfusion, and
immunomodulation in the critically ill. Chest. 2005; 127: 295-307.
- Xu XH, Yu XR, Huang YG. [Association of Perioperative Allogeneic
Red Blood Cell Transfusion with Postoperative Infections]. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2022; 44: 497-503.
- Wang W, Zhao L, Niu P, et al. Effects of perioperative blood transfusion in gastric cancer patients undergoing gastrectomy: A systematic review and meta-analysis. Front Surg. 2022; 9: 1011005.
- McSorley ST, Tham A, Dolan RD, et al. Perioperative Blood Transfusion is Associated with Postoperative Systemic Inflammatory
Response and Poorer Outcomes Following Surgery for Colorectal
Cancer. Ann Surg Oncol. 2020; 27: 833-43.
- Liu X, Ma M, Huang H, et al. Effect of perioperative blood transfusion on prognosis of patients with gastric cancer: A retrospective
analysis of a single center database. BMC Cancer. 2018; 18: 649.