Introduction
Traumatic Brain Injury (TBI) is a significant global burden in
terms of mortality and disability. TBI is a significant global burden
in terms of mortality and disability. TBI is a type of brain injury
caused by motor vehicle accidents, falls, violence, sports activities, and other factors. TBIs can be classified into different types,
including skull fractures, cerebral concussions, subarachnoid
hemorrhages, subdural hematoma, epidural hematoma, and cerebral contusion. The severity of TBI is often determined using the
Glasgow Coma Scale (GCS), which classifies TBIs as mild (GCS 13-15), moderate (GCS 8-12), or severe (GCS 3-7) [1]. Furthermore,
very severe TBI is defined as GCS 3-5. [2]. The all TBI cases are
mostly classified as mild (GCS 13-15), while approximately 0.6%
are severe TBI (GCS 3-7) and 0.3 % are very severe TBI [2]. Regarding recovery outcomes, approximately 8% of severe TBI cases achieve good recovery (Glasgow Outcome Scale, GOS 5), while
good recovery from very severe TBI (GCS 3-5 ) is rare [3]. Furthermore, good recovery from severe TBI with bilateral traumatic hematomas rages from 0 to 3% [4], while good recovery from very
severe TBI with bilateral intracranial hematomas is extremely rare.
Optimal treatment for bilateral traumatic intracranial hematomas remain largely unknown. When it comes to surgical treatment for bilateral traumatic intracranial hematomas, there is still
controversy regarding the most appropriate approach. The options under debate include bilateral craniectomies, bilateral craniotomies, unilateral craniectomy, or unilateral craniotomy [5,6].
Furthermore, there is a scarcity of detailed reports on the course
of very severe TBI cases with excellent outcomes. In this study, we
present a case of a very severe TBI with an excellent outcome following evacuation and craniectomy for acute epidural hematoma
and contra-lateral subdural hematoma associated with intracerebral contusional hemorrhage. Subsequently, we discuss the optimal treatment options for similar cases.
Case presentation
A 46-year-old man experienced a fall from a height of 2 meters
while at work, resulting in a blow to his left temporal head. He
was promptly transported to our hospital via emergency car. A CT
scan revealed the presence of a left temporal epidural hematoma
accompanied by a temporal bone fracture, as well as a contralateral acute subdural hematoma and contusional hemorrhage in
the temporal and frontal lobes. Upon admission, his level of consciousness was assessed using the Glasgow Coma Scale (GCS) and
scored 13 points. However, within two hours, his consciousness
rapidly declined to a GCS score of 4 points. Bilateral light reflexes
became significantly diminished and dilated, with the pupils exhibiting dilation and slight anisocoria (right>left). A follow-up CT
scan indicated the enlargement of bilateral hematomas, prompting the decision to proceed with emergency surgery. The initial
step involved performing a right temporal craniectomy, during
which a remarkably thin right temporal bone with a thickness of
1.8 mm was removed. Subsequently, durotomy was conducted,
and subdural and subcortical hematomas were evacuated to the
maximum extent possible. Following hematoma evacuation, a
Gore-Tex seat was placed on the brain surface without dura mater plasty, while the removed skull bone was positioned beneath
the abdominal subcutaneous tissues. In the subsequent stage,
a left temporal craniotomy was performed to fully evacuate the
epidural hematoma. The left temporal bone was also found to be
exceptionally thin, measuring 1.8 mm in thickness. Following the
operation, upon the patient’s return to the intensive care unit,
the pupils exhibited isocoria and the light reflexes were prompt.
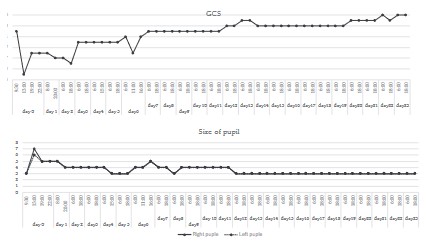
The GCS score improved to 8. postoperative CT scans confirmed
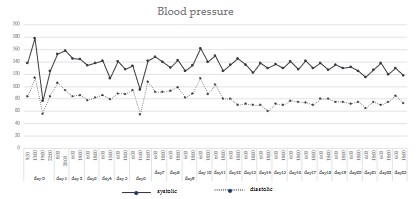
decompression on both sides of the brain. The episodic hypotension (<90 mHg) was recognized and dopamine was administrated
(Figure 1). Although left brain swelling resolved after the surgery,
right brain swelling persisted, and the patient’s GCS remained at
a low level between 6-8 points. On day 3 after the injury, the patient developed left hemiparesis in the upper and lower limbs. CT
scans indicated an increase in right brain swelling. The patient’s
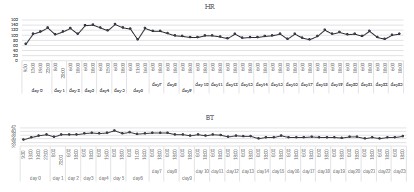
body temperature exceeded 39°C, and acetaminophen was administered to reduce the fever (Figure 2). On day 4 after the injury,
the patient experienced acute respiratory distress due to airway
narrowing. Oxygen saturation in the blood dropped below 85%, and arterial blood gas analysis revealed decreased O2 pressure
and increased CO2 pressure. Subsequently, trans-oral tracheal intubation was performed with spontaneous respiration. However,
hyper-ventilation exceeding 25 breaths per minute and tachycardia over 120 beats per minute persisted. Oxygen saturation in the
blood occasionally decreased. Consequently, on day 6 after the
injury, the patient was transitioned to artificial ventilation using
an artificial ventilator with synchronized intermittent mandatory
ventilation (SIMV) mode. This intervention improved the patient’s
respiratory status, with blood oxygen saturation exceeding 98%.
The pupils exhibited isocoria, and the light reflex became prompt
(Figure 3). However, the level of consciousness remained at a low
level between GCS 8 and 9. Intravenous infusion of glycerol at a
rate of 800 mL/day, prednisolone at a dose of 20 mg/day, and
flocemide at a dose of 20 mg/day were continued. On day 9 after
the injury, the ventilation mode was changed from SIMV mode to
continuous positive airway pressure (CPAP) mode. On day 11 after
the injury, the tracheal tube was removed, and oxygen was supplied at a rate of 3 L/minute via a facial mask. The patient maintained a blood oxygen saturation level of over 96%. The patient
responded to commands, opened his eyes, and moved his hands
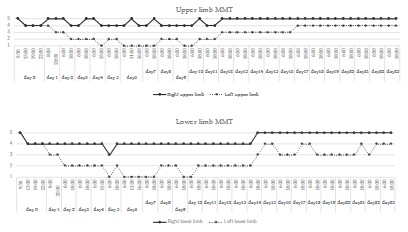
and legs, albeit with left hemiparesis (MMT: upper limb 3/5, lower
limb 4/5).
From day 12 to 14 after the injury, the patient’s condition remained relatively stable. The level of consciousness ranged between GCS 10-11, and left motor weakness persisted (MMT: up-
per limb 3/5, lower limb 4/5). On day 15 after the injury, the level
of consciousness improved to GCS 12, and the left motor weakness showed improvement (MMT: upper limb 4/5, lower limb
4/5) (Figure 4). Oxygen supply was discontinued as blood oxygen
saturation reached 98%. The patient began receiving soft foods
due to mild swallowing difficulties. On day 18 after the injury, the
level of consciousness further improved to GCS 13. On day 21 after the injury, the level of consciousness reached GCS 14, and left
hemiparesis improved to MMT (upper limb 4/5, lower limb 4/5).
The patient was able to stand and walk with the support of parallel bars. A follow-up head CT scan indicated a reduction in swelling
in the right hemisphere and the disappearance of the intracerebral hematoma. On day 24 after the injury, the level of consciousness reached GCS 15, and the left motor weakness disappeared
(Figure 4). On day 28 after the injury, the patient contracted COVID-19 and was quarantined in the hospital for 10 days. After the
COVID-19 quarantine period, the patient continued to experience
further improvement in higher brain function and limb functions.
On day 41 after the injury, an electroencephalography showed occasional paroxysmal discharges in the right temporal lobe. Another head CT scan demonstrated further reduction in swelling in the
right hemisphere and slight atrophy of that hemisphere (Figure 5). At this stage, the patient had fully recovered but required rehabilitation to address disuse-related issues. On day 63 after the
injury, the patient underwent cranioplasty using his own temporal bone, which was buried under the abdominal skin. By 70 day
after the injury, mild recent memory disturbance and attention
disorder were observed in the patient’s higher brain functions.
On day 76 after the injury, the patient was discharged home. On
day 132 after hospital discharge, the patient underwent an ophthalmological examination due to visual disturbances, which revealed bilateral upper left quarter anopsia (Figure 6). By day 140
after the injury, the patient had completely returned to work.
Written informed consent for publication of the research details and clinical images were obtained from the patient. The patient provided informed consent after the study. This study was
approved by the Institutional Review Board of Asahi Hospital (No. 2023-1).
Discussion
Traumatic acute extra-axial hematomas, including acute epidural hematomas (AEDHs) and acute subdural hematomas (ASDHs),
often occur in conjunction with intracerebral contusional hemorrhage. AEDHs typically result from a direct blow to the temporal
region, occasionally causing a skull fracture and subsequent rupture of the middle meningeal artery. These hematomas rapidly increase in size and can lead to herniation syndromes once a criti-
cal intracranial pressure threshold is reached [7]. In some cases,
AEDHs can cause contralateral subdural hematomas and contusions, as observed in the present case. On the other hand, ASDHs
commonly result from deceleration forces that cause the brain's
surface to shear against the undersurface of the skull, leading to
injury of the bridging veins [8].
Currently, guidelines for the surgical management of traumatic
brain injury recommend the following [9]. Surgical evacuation for
AEDHs should be performed for hematomas larger than 30 cm3,
irrespective of the Glasgow Coma Scale (GCS) score. However, AEDHs measuring less than 30 cm3 with a GCS score greater than 8
can be managed without surgery, with close neurological observation. Similarly, surgical evacuation for ASDHs is recommended
for patients with a hematoma thickness greater than 10 mm or
midline shift exceeding 5 mm if the GCS score rapidly declines to
less than 9 and the pupils show asymmetry or dilation. Craniotomy or craniectomy with bone flap removal and dura mater plasty
are the preferred surgical techniques for evacuation. Furthermore, surgical intervention for traumatic contusional parenchymal hemorrhage (TCPH) is recommended for patients exhibiting
progressive neurological deterioration and refractory intracranial
hypertension. Additionally, patients with parenchymal hemorrhage larger than 50 cm3 are recommended to undergo operative treatment. Conversely, patients without neurological deficits
and no significant signs of mass effect can be managed without
surgery but require intensive monitoring and serial imaging. Regarding bilateral traumatic intracranial hematoma, the surgical
indications are as follows: 1) signs of brain herniation; 2) clinical
deterioration of consciousness with hemorrhagic progression; 3)
evident mass-occupying effect and compression of basal cisterns.
The decision on whether to perform unilateral or bilateral surgery
should be based on clinical, radiological, and ICP findings, with
neurosurgeons making the final determination. Surgical operations can be performed simultaneously or separately if both lesions require surgical intervention [5,6].
In the present case, a coup epidural hematoma, contralateral
subdural hematoma, and contusional parenchymal hemorrhage
were observed. Bilateral craniotomies with unilateral bone flap
removal were performed to address the bilateral intracranial hematomas. In addition, the patient exhibited cerebral edema, re-
spiratory dysfunction, transient hypotension, anemia, infection,
hyperthermia, tachycardia with mild heart failure, hyperglycemia,
pulmonary infection, and liver dysfunction. The patient was mechanically ventilated and underwent tracheal intubation in the
Neurological Intensive Care Unit (NICU). Several guidelines for
traumatic brain injury provide recommendations for managing
these conditions [10-13].
Transient hypotension (systolic blood pressure <90 mmHg)
lasting at least 5 minutes has been associated with a significant
increase in neurological morbidity and mortality. Therefore, blood
pressure should be closely monitored, and hypotension should be
avoided. Rapid correction of hypotension can be achieved using
vasopressor drugs such as phenylephrine, norepinephrine, and
catecholamines. The duration of hypoxemic episodes (< SaO2 90%)
is a crucial predictor of mortality. Oxygenation levels should be
monitored, and hypoxia (PaO2 < 60 mmHg or O2 saturation <90%)
should be prevented [10]. Stress-related hyperglycemia in TBI is induced by counter-regulatory hormones and/or insulin resistance,
further damaging the injured brain tissue. Hyperglycemia after
TBI is associated with an increased risk of mortality and poor neurological outcomes. Therefore, maintaining serum glucose concentrations between 8 mmol/L (1.4 g/L) and 10-11 mmol/L (1.8-2
g/L) is recommended for patients with severe TBI [11]. To prevent
infection with intubation, administration of peri-procedural antibiotics is recommended to reduce the incidence of pneumonia.
Early tracheostomy should be considered to reduce the duration
of mechanical ventilation. However, for antiseizure prophylaxis,
the routine use of phenytoin or valproate is not recommended
for preventing late posttraumatic seizures (PTS). Hyperventilation
is recommended as a temporary measure to reduce elevated intracranial pressure (ICP), but prophylactic hyperventilation (PaCO2
of 25 mmHg or less) is not recommended [12]. Osmotherapy is
commonly employed to control cerebral edema. Hypertonic solutions, such as mannitol and glycerol, are used in osmotherapy.
It is recommended to administer mannitol 20%, glycerol 10%, or
hypertonic saline solution (at 250 mOsm) via infusion over 15-20
minutes to manage threatened intracranial hypertension or signs
of brain herniation. Compared to mannitol, glycerol has similar
effectiveness. Glycerol is associated with a significantly lower risk
of acute kidney injury, electrolyte disturbances, and rebound effects [13]. On the other hand, high-dose glucosteroids are not recommended for improving outcomes or reducing ICP. High-dose
methylprednisolone is associated with increased mortality and
is contraindicated [9]. We presented an exceptional outcome in
a case of very severe TBI with a GCS score of 4. The complete
recovery in this case is likely attributed not only to surgical treatment but also to systemic management, including therapies for
hyperthermia, hyperglycemia, respiratory dysfunction, and episodic hypotension. Hyperthermia, hyperglycemia, and respiratory
dysfunction were speculated to be associated with brain edema
affecting the hypothalamus and midbrain. These pathological
conditions are interconnected, emphasizing the importance of integrated medical treatment in the ICU. In this case, glycerol 10%
was used for two weeks to reduce brain edema. Although mannitol 20% can rapidly reduce brain edema, it can sometimes lead to
acute renal failure. Therefore, complications associated with the
use of glycerol were not observed. Dopamine hydrochloride was
used for episodic hypotension, sliding-scale insulin for hyperglycemia, tracheal intubation and the supply of high-concentration
oxygen for respiratory dysfunction and decreased blood oxygen
saturation, followed by mechanical ventilation. Antibiotics were
administered to control respiratory system infections. Body cooling and acetaminophen were employed to manage hyperthermia.
The combination of monoammonium glycyrrhizinate and cysteine
hydrochloride (MG-CH) was used to treat liver dysfunction [14].
The patient contracted COVID-19 on day 27 after the injury and
received a 5-day course of the anti-SARS-CoV-2 agent Molnupiravir without developing respiratory symptoms. Cognitive impairments have been observed in patients recovering from COVID-19
[15]. However, the patient did not show cognitive deterioration
after COVID19. The absence of cognitive deterioration may be
attributed to either being in the postinjury convalescent period
during the COVID-19 infection or the lack of significant changes
due to the effect of Molnupiravir. The patient's motor and cognitive functions had almost fully recovered by the time of the cranioplasty operation. At discharge, the patient exhibited only left
quadrant hemianopsia. This rare case of a patient with very severe TBI achieved an excellent outcome and was able to return to
work. We provided appropriate treatment to the patient, closely
adhering to the guidelines for severe TBI. It is crucial to thoroughly understand the individual conditions of patients with severe TBI
and provide them with the appropriate treatments. Without such
comprehensive and tailored care, patients with severe TBI may
not be able to be saved or recover without disability.
Conclusion
In conclusion, we presented an extremely rare case of a patient
who achieved an excellent outcome following a very severe traumatic brain injury, which included an acute epidural hematoma,
contralateral subdural hematoma, and intracerebral contusional
hemorrhage. Despite the high mortality and morbidity associated
with such cases, it is possible to rescue some patients, and rare
cases may achieve full recovery if the individual conditions of patients with severe TBI are thoroughly understood and appropriate
treatments are provided.
Declarations
Ethical statement: This study was approved by the Institutional Review Board of Asahi Hospital (No. 2023-1). The patient provided informed consent after the study. Written informed consent
for publication of the research details and clinical images were
obtained from the patient.
Acknowledgement: We thank the nursing team and the rehabilitation staffs of Asahi Hospital and the patient for allowing us to
publish the treatment outcome.
Conflicts of interest: The authors have no conflicting financial
interest.
References
- Stocchetti N, Carbonara M, Citerio G, Ercole A, Skrifvars MB, et al.
Severe traumatic brain injury: targeted management in the intensive care unit. Lancet Neurol. 2017; 16: 452-464.
- Van Dijck JT, Reith FC, van Erp IA, et al. Decision making in very severe traumatic brain injury (Glasgow Coma Scale 3-5): a literature
review of acute neurosurgical management. J Neurosurg Sci. 2018;
62: 153-177.
- Van Dijck JTJM, Mostert CQB, Greeven APA, et al. Functional outcome, in-hospital healthcare consumption and in-hospital costs for
hospitalised traumatic brain injury patients: a Dutch prospective
multicentre study. Acta Neurochir (Wien). 2020; 162: 16071618.
- Pandey S, Sharma V, Singh K, et al. Bilateral Traumatic Intracranial
Hematomas and its Outcome: a Retrospective Study. Indian J Surg.
2017; 79: 19-23.
- Hu Y, Sun H, Yuan Y, Li Q, et al. Acute bilateral mass-occupying lesions in non-penetrating traumatic brain injury: a retrospective
study. BMC Surgery. 2015; 15: 6.
- Pandey S, Sharma V, Singh K, et al. Bilateral traumatic intracranial
hematomas and its outcome: a retrospective study. Indian J Surg.
79: 19-23.
- Bullock MR, Chesnut R, Ghajar J, et al. Surgical management of
Traumatic Brain Injury Author Group. Surgical management of
acute epidural hematomas. Neurosurgery. 2006; 58(3): S7-15.
- Karibe H, Hayashi T, Hirano T, Kameyama M, Nakagawa A, Tominaga T. Surgical management of traumatic acute subdural hematoma
in adults: a review. Neurol Med Chir. 2014; 54: 887-894.
- Hawryluk GWJ, Rubiano AM, Totten AM, et al. Guidelines for the
management of severe traumatic brain injury: 2020 Update of the
decompressive craniectomy recommendations. Neurosurgery.
2020; 87: 427-434.
- The Brain Trauma Foundation. The American Association of Neurological Surgeons. The Joint Section on Neurotrauma and Critical Care. Resuscitation of blood pressure and oxygenation. J Neurotrauma. 2000; 17(6-7): 471-478.
- Shi J, Dong B, Mao Y, Guan W, Cao J, Zhu R, Wang S. Review: Traumatic brain injury and hyperglycemia, a potentially modifiable risk
factor. Oncotarget. 2016; 7: 71052-1061.
- Carney N, Totten AM, O’Reilly C, et al. Guidelines for the Management of Severe Traumatic Brain Injury, Fourth Edition. Neurosurgery. 2017; 80: 6-15.
- Wang J, Ren Y, Zhou LJ, Kan LD, Fan H, Fang HM. Glycerol infusion versus mannitol for cerebral edema: A systematic review and
meta-analysis. Clin Ther. 2021; 43: 637e649.
- Chu S, Niu Z, Guo Q, et al. Combination of monoammonium glycyrrhizinate and cysteine hydrochloride ameliorated lipopolysaccharide/galactosamine-induced acute liver injury through Nrf2/ARE
pathway. Eur J Pharmacol. 2020; 882: 173258.
- Tavares-Júnior JWL, de Souza ACC, Borges JWP, Oliveira DN, Siqueira-Neto JI, Sobreira-Neto MA, Braga-Neto P. COVID-19 associated
cognitive impairment: A systematic review. Cortex. 2022; 152: 77-97.