Introduction
Nipple discharge (ND) is the third most common complaint in
patients with breast diseases after breast pain and breast mass
and is mainly seen in women aged 30-50 [1,3]. Physiologic nipple
discharge is usually milky or water-like, spontaneous, and multiductal. In contrast, pathological nipple discharge (PND) is bloody
or serous-like, spontaneous, unilateral, and single-holed [3,4].
Intraductal papilloma (IDP) is the most common cause of PND,
with an occurrence of 63-77% [5,6]. However, 5-15% of patients
with PND are diagnosed with malignant ductal carcinoma in situ
and breast cancer [7,10]. According to the guideline of UpToDate,
most PND needs surgical management. Currently, conventional
examinations for diagnosing and locating the cause of PND mainly
include mammary fiberoptic ductoscopy, galactography, ND cytology, and ultrasonography. However, each of these modalities
has certain limitations [11,12]. Limited by the outer optical fiber
diameter and different mammary duct branching modes, the observation range of ductoscopy is mainly confined to the central
mammary duct [13]. Galactography is not recommended because
of its high technical failure rate, invasive nature, and false-negative rate [14]. Recently, microbubble-based contrast agents have
been manufactured for contrast-enhanced ultrasound (CEUS) to
diagnose breast diseases [15,16]. These enhanced agents reflect
more echogenic signals after intravenous injection to determine
the position and features of the blood vessels. However, the application of this kind of research involving injecting microbubblebased contrast agents into the mammary duct for clear visualization has not yet been reported in PND patients. The Vacuum-assisted breast biopsy (VABB), also known as mammotome® biopsy,
is a novel device used extensively in Japan, Europe, and America
[17,19]. It is a minimally invasive approach to breast puncture for
the qualitative diagnosis before breast cancer surgery and evaluation of breast microcalcifications and papillary lesions. For the accuracy of preoperative localization and minimally invasive results,
this study combined the advantages of ultrasound to investigate
the value of breast duct CEUS and minimally invasive duct excision using microbubble-based contrast agents combined with the
Mammotome system in PND patients.
Methods
Patients
This study was approved by The Medical Ethics Committee
of Beijing Friendship Hospital, Capital Medical University, and it
is registered at ClinicalTrials.gov.cn (ID: ChiCTR1900022776). All
participants were provided with written informed consent before
enrollment in this study between May 2019 and May 2020. In addition, all PND patients registered in the Department of General
Surgery were subjected to qualification screening. The inclusion
criteria of this study were as follows: (1) underwent ultrasoundguided breast-focused minimally invasive resection using the
Mammotome system; (2) subjected to preoperative breast duct
CEUS; (3) age>18 years; and (4) voluntarily enrolled in this study
and provided written informed consent. The exclusion criteria
were as follows: (1) definitive diagnosis of breast cancer with
puncture pathology or ND cytology; (2) history of malignant tumors of the breast or other organs; and (3) poor general physical
condition, incapable of undergoing an operation.
Instruments and materials: The instruments used in our study included the iU22 color Doppler ultrasound system with a superficial L9-3 linear transducer for contrast-enhanced sonography
(Philips Healthcare, USA), 8-gauge Mammotome breast biopsy
system (Devicor Medical Products, USA), and PD-VC-0210 endoscope camera system of the mammary duct (Ethicon Endo-surgery, USA). In addition, SonoVue (Bracco Suisse SA, Switzerland)
sulfur hexafluoride microbubbles were used for injection.
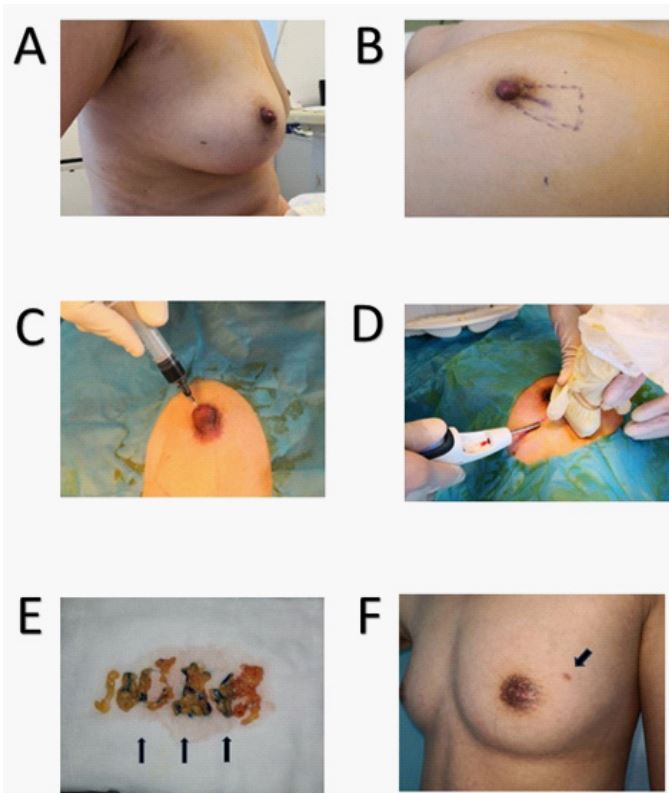
Surgical procedures: Routine ultrasound examination. The patient was placed in the supine position. Ultrasonography examination of the PND patients included a review of breast tissue, dilated mammary ducts, hypoechoic nodules, and bilateral axillary
lymph nodes. In addition, clear and comprehensive ultrasound
images were selected and stored for comparison with the CEUS
breast duct images.
Ductoscopy: The nipple-areola complex was cleaned with a
povidone-iodine solution, and ductoscopy was performed under local anesthesia using diluted lidocaine (0.5%). First, a rude
pinhead was placed in the dilated ductal orifice. The expander
system was then introduced into the ductal orifice to expand the
duct gently to submit the fiberoptic scope. A ductoscopy examination is done in a regular way and is well documented [20-22].
Finally, a soft syringe needle was inserted into the dilated ductal
orifice for further injection.
Breast duct CEUS: Breast duct CEUS was conducted to monitor
the branching and pathway of the mammary duct. First, physiological saline (5 ml) was mixed with SonoVue lyophilization and
vigorously shaken for 20 seconds to obtain the SonoVue suspension. Next, 1-4 ml of the configured SonoVue suspension was injected into the mammary duct through the soft syringe needle
into the dilated ductal orifice. The pathway of the mammary duct
was visualized immediately on the contrast pulse sequence after
injection. The contrast ultrasound can help surgeons identify the
scope of the mammary duct lesions to achieve complete duct resection.
Results
Clinical and demographic characteristics: This study enrolled 20
patients with PND who underwent preoperative breast duct CEUS
and Mammotome‐assisted minimally invasive resection between
May 2019 and May 2020. Table 1 shows the clinical and demographic characteristics of the patients. All patients were women,
and the average age was 47.5 years. Notably, the occurance of the
right- and left-sided lesions was the same. Regarding the pathological classification, 14 patients were diagnosed with IDP, 5 with
duct ectasia, and 1 with intraductal papillary carcinoma. Of the 20
patients, 7 did not show positive findings for lesions on ductoscopy, and only 1 did not show positive results on duct CEUS.
Postoperative effect and patient satisfaction: The follow-up period ranged from 12 to 24 months (average, 18±6 months).
Postoperative evaluation was performed by the patients and the
surgeon at each follow-up visit. Patient satisfaction, recurrence,
symmetry, infection, and scarring were evaluated. There was no
recurrence reported in any patient. The bilateral breast tissue was
assessed during follow-up using ultrasound. None of the patients
experienced severe postoperative complications, and all were
very satisfied with the results. Incision scars were nearly invisible [6-8] months after the procedure. All patients recovered well
without infection and were very satisfied with the results.
Table 1: Clinical and demographic characteristics of patients.
| Characteristics |
Total |
| Sex |
|
| Female, n(%) |
20(100 %) |
|
Age (mean ± standard
deviation)
|
47.50±12.95 years |
| Lesion location |
|
| Left, n(%) |
10(50%) |
| Right, n(%) |
10(50%) |
|
Pathological classification
|
|
|
Intraductal papilloma, n(%)
|
14(70%) |
| Duct ectasia, n(%) |
5(25%) |
|
Intraductal papillary
carcinoma, n(%)
|
1(5%) |
| Ductoscopy |
|
| Positive, n(%) |
13(65%) |
|
Duct contrast-enhanced
ultrasound
|
|
| Positive, n(%) |
19(95%) |
Discussion
ND is classified as pathologic if it is spontaneous, unilateral, or
bloody. Alternatively, serous, and clear discharge is usually associated with a tumor [12]. IDP, duct ectasia, carcinoma, and infection
are common causes of PND. Benign papilloma is the most common cause of these diseases, identified in up to 57% of cases presenting PND [23]. Currently, conventional examinations for diagnosing the cause of PND include mammary fiberoptic ductoscopy,
galactography, ND cytology, and ultrasonography. However, each
of these has certain limitations. Limited by the outer optical fiber
diameter and different mammary duct branching modes, the observation range of ductoscopy is mainly confined to the central
mammary duct [23]. Although mammography is recommended
for all women with PND over 30 years, the sensitivity (7-68%) for
detecting malignancy associated with PND is not sufficient and
satisfactory [24]. Mammary ductoscopy has been used for over
40 years since its introduction in 1988 to evaluate spontaneous
and bloody ND patients. Ductoscopy is a minimally invasive procedure that helps in the visualization of the ductal epithelium of
the breast via the nipple. Although some studies [25,26] have
shown a strong correlation between intraductal morphological
images and histopathologic diagnosis, others have shown that
this relationship is only valid for papillomas [22,27,28]. Ductoscopy provides good visualization of approximately 94% of lesions,
the specificity for malignancy it’s much higher than benign lesions
[20]. These results indicate that histological diagnosis or surgery is necessary to exclude malignancy in patients with PND [20]. The
primary indications for mammary ductoscopy are to evaluate PND
and assess the risk of developing breast cancer and other diseases
[29]. Using an endoscope with a 1.0 mm outer diameter may not
allow the detection of lesions in the margin of the breast. In other words, we concluded that false-negative results might mainly
be due to the possibility of lesion development from the distal
ductal units. Peripheral distribution of lesions results in a falsenegative rate of 18% with ductoscopy. Additionally, galactography
is contraindicated in pregnant women because of the radiationinduced damage. As for ND cytology, its low sensitivity makes it
difficult to improve the detection rate; therefore, only positive
results in ND cytology are considered clinically significant [30]. Ultrasonography provides good resolution for hypoechoic nodules
in the breast. However, PND patients often have multiple small
hypoechoic nodules, making it challenging to identify the nature
of the hypoechoic areas with ultrasound [31]. Therefore, a more
precise preoperative examination to determine the scope of the
mammary duct is highly desirable.
To overcome the limitations of ductoscopy, we suggest that all
PND cases be subjected to breast duct CEUS, a noninvasive technique that can solve the above problem [32]. The new generation
of ultrasound contrast agents, in combination with low acoustic
power contrast-specific ultrasound imaging, has enabled a major
clinical breakthrough in some organs’ diagnostic and interventional procedures [33,34]. In China, the only licensed contrast agent is
SonoVue, a sulfur hexafluoride-filled microbubble contrast agent
stabilized by phospholipids. CEUS plays a crucial role in evaluating breast lesions by the intravenous injection of contrast agents.
However, there are few reports of the use of CEUS in diagnosing
intraductal lesions and localizing them after SonoVue injection in
the mammary duct in clinical settings.
Conventionally, the standard approach in PND cases is to perform duct excision, which involves complete excision of the corresponding mammary ducts and terminal ducts. A better approach
is to inject a mixture of methylene blue or an opaque dye and
perform preoperative breast angiography that can show the duct
progression and assist in intraoperative identification of the target ducts, which will be stained in blue dye. These surgical procedures can help detect possible premalignant lesions and carcinomas. This surgical procedure is relatively straightforward and has
minimal morbidity. However, regardless of the type of traditional
surgery, an incision of at least 2-3 cm is required on the breast
surface, and a surgical scar is unavoidable.
Minimally invasive mastectomy, also called ultrasound-guided
vacuum-assisted breast biopsy system, has been used to remove
breast nodules since 1995. The Mammotome biopsy system is a
complete breast disease sampling and management system. A
Mammotome 8-gauge needle can yield a cylinder-shaped tissue
sample measuring approximately 23 mm in length and 4.3 mm
in diameter per cut, allowing for quick and precise duct and glandular tissue excision [35]. These excisions are usually limited to
lesions less than 30 mm, mainly due to time, patient comfort, and
tolerance. It has also been reported to be helpful in male breast
development surgery [36]. However, there are only a few reports
on this procedure’s use in managing PND. The minimally invasive
operations for ND have a shorter surgical time, but it is not significantly different from the traditional method. The complications of this technique include bleeding and hematomas that result from
sharp dissection; these complications can be minimized by using
the tumescent anesthesia technique and an intraoperative Doppler ultrasound to prevent cutting the large vessels. An incision
only 5 mm long is considered the reason for esthetic satisfaction
in all patients. After at least 1 year of follow-up, the result of this
minimally invasive surgery seemed satisfactory in almost all patients.
Some risks are associated with this type of surgery, including
the inability to breastfeed in younger patients, loss of nipple sensation, and possible nipple-areolar necrosis. Therefore, we believe
all intraductal lesions detected on ultrasound ductoscopy, CEUS,
or magnetic resonance imaging should be subjected to minimally
invasive duct resection.
Limitations: This study has a few limitations. First, it was challenging to convince all the enrolled patients to undergo an ultrasound, magnetic resonance imaging, CEUS, and ductoscopy because of time constraints and high costs. Second, the number of
breast PND cases was limited. Third, it is not easy to perform CEUS
through the discharging duct, and a surgeon needs the full cooperation of the ultrasonography technician. Furthermore, there is a
specific learning curve involved. Additionally, a larger sample size
and a more extended follow-up period will be required to determine the long-term safety and efficacy of this minimally invasive
surgical technique.
Conclusions
Combining conventional ultrasound, CEUS, and ductoscopy
has an obvious advantage in preoperative localization and surgical treatment of patients with PND. In current clinical practice,
CEUS is necessary to compensate for the limitations of ductoscopy when evaluating patients with PND. All patients who underwent vacuum-assisted breast excision reported sound therapeutic effects and cosmetic outcomes, which avoided open surgical
procedures and huge scars. Minimally invasive surgery, generally
well tolerated by patients, may be a safe and efficient procedure
for managing breast PND with or without lesions. Moreover, the
complication rate was acceptable.
Declarations
Acknowledgments: We are very grateful to the team for their
guidance and help in diagnosing and treating. Thank the patients
for their understanding and cooperation in this study. Thank the
nursing team for their efforts in the treatment process.
Authors’ contributions: (I) Conception and design: YG Gao; (II)
Administrative support: ZC Ge; (III) Provision of study materials or
patients: YG Gao, ZH Wang, HM Zhang; (IV) Collection and assembly data: ZY Yang, YG Gao; (V) Data analysis and interpretation: YG
Gao, ZY Yang; (VI) Manuscript writing: YG Gao, ZY Yang; (VII) Final
approval of manuscript: All authors.
Funding: No foundation supports this work.
Availability of data and materials: The data will not be made
available to protect the participant’s identity. However, upon
reasonable request, those interested parties can contact the corresponding author Dr. Yinguang Gao (gaoyinguang@gmail.com),
who will provide the raw data.
Ethics approval and consent to participate: All procedures in
this study involving human participants were by the Declaration
of Helsinki (as revised in 2013). This study was approved and supervised by the Medical Ethics Committee of Beijing Friendship
Hospital, Capital Medical University. (NO.2019-P2-062-01). All
participants provided informed consent.
Consent for publication: Not applicable.
Competing interests: The authors declare that they have no
competing interests.
References
- Bodine AM, Holahan B and Mixon A. Benign Breast Conditions. J Am Osteopath Assoc. 2017; 117: 755-760. 2017/11/29. DOI: 10.7556/jaoa.2017.147.
- Nurudeen S. Breast Health: Benign Breast Conditions. FP Essent. 2020; 496: 16-20. 2020/09/10.
- Roman M, Louro J, Posso M, et al. Breast density, benign breast disease, and risk of breast cancer over time. Eur Radiol. 2021; 31: 4839-4847. 2021/01/08. DOI: 10.1007/s00330-020-07490-5.
- Pleasant V. Benign Breast Disease. Clin Obstet Gynecol. 2022; 65: 448-460. 2022/06/17. DOI: 10.1097/GRF.0000000000000719.
- Rodden AM. Common breast concerns. Prim Care. 2009; 36: 103-113, viii. 2009/02/24. DOI: 10.1016/j.pop.2008.10.006.
- Pruthi S. Detection and evaluation of a palpable breast mass. Mayo Clin Proc. 2001; 76: 641-647; quiz 647-648. 2001/06/08. DOI: 10.4065/76.6.641.
- Santen RJ and Mansel R. Benign breast disorders. N Engl J Med. 2005; 353: 275-285. 2005/07/22. DOI: 10.1056/NEJMra035692.
- Miltenburg DM and Speights VO, Jr. Benign breast disease. Obstet Gynecol Clin North Am. 2008; 35: 285-300, ix. 2008/05/20. DOI: 10.1016/j.ogc.2008.03.008.
- Beca F, Oh H, Collins LC, et al. The impact of mammographic screening on the subsequent breast cancer risk associated with biopsy-proven benign breast disease. NPJ Breast Cancer. 2021; 7: 23. 2021/03/07. DOI: 10.1038/s41523-021-00225-9.
- Tran WT, Sadeghi-Naini A, Lu FI, et al. Computational Radiology in Breast Cancer Screening and Diagnosis Using Artificial Intelligence. Can Assoc Radiol J. 2021; 72: 98-108. 2020/09/01. DOI: 10.1177/0846537120949974.
- Bevers TB, Helvie M, Bonaccio E, et al. Breast Cancer Screening and Diagnosis, Version 3.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018; 16: 1362-1389. 2018/11/18. DOI: 10.6004/jnccn.2018.0083.
- Filipe MD, Simons JM, Moeliker L, et al. Patient-reported outcomes of ductoscopy procedures for pathologic nipple discharge. Breast Cancer. 2021; 28: 471-477. 2020/11/13. DOI: 10.1007/s12282-020-01184-y.
- Chang YK, Chen CT, Wang M, et al. Could ductoscopy alleviate the need of microdochectomy in pathological nipple discharge? Breast Cancer. 2020; 27: 607-612. 2020/02/03. DOI: 10.1007/s12282-020-01051-w.
- Waaijer L and Witkamp AJ. Management of Nipple Discharge and the Associated Imaging Findings: Comments to the Editor. Am J Med. 2015; 128: e29. 2015/09/01. DOI: 10.1016/j.amjmed.2015.01.018.
- Esfehani MH, Yazdankhah-Kenari A, Omranipour R, et al. Validation of Contrast Enhanced Ultrasound Technique to Wire Localization of Sentinel Lymph Node in Patients with Early Breast Cancer. Indian J Surg Oncol. 2015; 6: 370-373. 2016/04/12. DOI: 10.1007/s13193-015-0446-4.
- Cao XL, Bao W, Zhu SG, et al. Contrast-enhanced ultrasound characteristics of breast cancer: correlation with prognostic factors. Ultrasound Med Biol. 2014; 40: 11-17. 2013/11/12. DOI: 10.1016/j.ultrasmedbio.2013.08.014.
- Ding B, Chen D, Li X, et al. Meta analysis of efficacy and safety between Mammotome vacuum-assisted breast biopsy and open excision for benign breast tumor. Gland Surg. 2013; 2: 69-79. 2013/05/01. DOI: 10.3978/j.issn.2227-684X.2013.05.06.
- Bozzini A, Cassano E, Raciti D, et al. Analysis of Efficacy and Accuracy of 2 Vacuum-Assisted Breast Biopsy Devices: Mammotome and Elite. Clin Breast Cancer. 2018; 18: e1277-e1282. 2018/08/04. DOI: 10.1016/j.clbc.2018.06.014.
- Pan S, Liu W, Jin K, et al. Ultrasound-guided vacuum-assisted breast biopsy using Mammotome biopsy system for detection of breast cancer: results from two high volume hospitals. Int J Clin Exp Med. 2014; 7: 239-246. 2014/02/01.
- Waaijer L, Simons JM, Borel Rinkes IH, et al. Systematic review and meta-analysis of the diagnostic accuracy of ductoscopy in patients with pathological nipple discharge. Br J Surg. 2016; 103: 632-643. 2016/03/24. DOI: 10.1002/bjs.10125.
- Okazaki A, Okazaki M, Asaishi K, et al. Fiberoptic ductoscopy of the breast: a new diagnostic procedure for nipple discharge. Jpn J Clin Oncol. 1991; 21: 188-193. 1991/06/01.
- Kamali S, Bender O, Aydin MT, et al. Ductoscopy in the evaluation and management of nipple discharge. Ann Surg Oncol. 2010; 17: 778-783. 2009/12/17. DOI: 10.1245/s10434-009-0820-y.
- Filipe MD, Patuleia SIS, de Jong VMT, et al. Network Meta-analysis for the Diagnostic Approach to Pathologic Nipple Discharge. Clin Breast Cancer. 2020; 20: e723-e748. 2020/07/16. DOI: 10.1016/j.clbc.2020.05.015.
- Motoda N, Ohashi R, Makino T, et al. Utility of duct-washing cytology for detection of early breast cancer in patients with pathological nipple discharge: A comparative study with fine-needle aspiration cytology. Diagn Cytopathol. 2020; 48: 1273-1281. 2020/08/09. DOI: 10.1002/dc.24572.
- Makita M, Akiyama F, Gomi N, et al. Endoscopic and histologic findings of intraductal lesions presenting with nipple discharge. Breast. J 2006; 12: S210-217. 2006/09/09. DOI: 10.1111/j.1075-122X.2006.00324.x.
- Denewer A, El-Etribi K, Nada N, et al. The role and limitations of mammary ductoscope in management of pathologic nipple discharge. Breast J. 2008; 14: 442-449. 2008/08/05. DOI: 10.1111/j.1524-4741.2008.00620.x.
- Louie LD, Crowe JP, Dawson AE, et al. Identification of breast cancer in patients with pathologic nipple discharge: does ductoscopy predict malignancy? The American journal of surgery. 2006; 192: 530-533.
- Dietz JR, Crowe JP, Grundfest S, et al. Directed duct excision by using mammary ductoscopy in patients with pathologic nipple discharge. Surgery. 2002; 132: 582-587; discussion 587-588. 2002/10/31. DOI: 10.1067/msy.2002.127672.
- Valdes EK, Boolbol SK, Cohen JM, et al. Clinical Experience With Mammary Ductoscopy. Ann Surg Oncol. 2016; 23: 9015-9019. 2006/08/10. DOI: 10.1245/ASO.2006.08.025.
- Ashfaq A, Senior D, Pockaj BA, et al. Validation study of a modern treatment algorithm for nipple discharge. Am J Surg. 2014; 208: 222-227. 2014/04/29. DOI: 10.1016/j.amjsurg.2013.12.035.
- Alcock C and Layer GT. Predicting occult malignancy in nipple discharge. ANZ J Surg 2010; 80: 646-649. 2010/09/16. DOI: 10.1111/j.1445-2197.2010.05270.x.
- SuteClaudon M, Cosgrove D, Albrecht T, et al. Guidelines and good clinical practice recommendations for contrast enhanced ultrasound (CEUS) - update 2008. Ultraschall Med. 2008; 29: 28-44. 2008/02/14. DOI: 10.1055/s-2007-963785.
- Dietrich CF, Nolsoe CP, Barr RG, et al. Guidelines and Good Clinical Practice Recommendations for Contrast Enhanced Ultrasound (CEUS) in the Liver - Update 2020 - WFUMB in Cooperation with EFSUMB, AFSUMB, AIUM, and FLAUS. Ultraschall Med. 2020; 41: 562-585. 2020/07/25. DOI: 10.1055/a-1177-0530.
- Tang X. Mammotome-Assisted Liposuction: A Novel Technique for Accessory Breasts. Aesthetic Plast Surg. 2017; 41: 517-523. 2017/01/08. DOI: 10.1007/s00266-016-0775-x.
- Atallah NG. [Ultrasound guided vacuum breast biopsy for microcalcifications in a male. First case report]. J Med Liban. 2006; 54: 161-163. 2006/12/28.