Introduction
Cerebrospinal Fluid (CSF) diversion is the main method of
treating hydrocephalus [1], with an estimated 30,000 ventriculostomies performed annually in the United States. The incidence
of Ventriculoperitoneal Shunt (VPS) failure during the first year
after initial implantation has been reported to be as high as 11-
25% [2,3]. VPS account for 97.7% of all implantations [4], but as
the number of implanted bypass systems in the abdominal cavity
increases, so does the number of complications, including intraventricular infections, bending or obstruction of the catheter, and
abdominal complications such as intestinal obstruction and traumatic injuries to intra-abdominal organs [5-8]. Infectious complications typically occur within the first 6 months after VPS, with
the majority occurring within the first 3 months after surgery. Late
infectious complications are rare and have different mechanisms.
The frequency of shunt infections, according to various sources,
ranges from 0.17% to 30% [9]. Peritonitis associated with VPS, as
a complication of meningitis, is mainly seen in pediatric patients
[10-12].
Case presentation
A 28-year-old woman was admitted to City Mariinsky Hospital
with complaints of headache, pain in the left and lower abdomen,
a body temperature of 38.5°C, severe weakness, and nausea. According to her medical history, she first experienced symptoms
of the disease 9 days prior to her hospitalization at City Mariinsky Hospital. These symptoms included catarrhal symptoms,
headache, a fever of 38.5°C, acute abdominal pain, nausea, and
repeated vomiting. She attempted to treat herself at home with
non-steroidal analgesics but did not experience significant relief. On the ninth day after experiencing first symptoms, she was
urgently hospitalized at the Clinical Infectious Diseases Hospital
named after S.P. Botkin with a suspected acute intestinal infection. However, after a comprehensive examination, this diagnosis
was ruled out. An ultrasound of her abdominal organs revealed a significant amount of fluid in the abdominal cavity. As a result, she
was transferred to City Mariinsky Hospital on the same day with a
suspected surgical pathology, specifically an “acute abdomen”. It
is also important to note that the patient has a history of perinatal
damage to her central nervous system in the form of intraventricular hemorrhage during childbirth. This condition was further
complicated by the development of occlusive hydrocephalus. The
patient has undergone ventriculoperitoneal shunting procedures
in 1995, 1997, 2003, and 2006. She currently works as a nurse and
denies any bad habits. Additionally, she denies any contact with
infectious patients.
Upon admission to the hospital, the patient’s condition was
serious. The neurological status was marked by drowsiness and
lethargy, with the patient answering questions correctly but only
in monosyllables. The patient’s body position was passive, lying
down. The skin appeared clean and of normal color, temperature,
and humidity, with no rashes, hemorrhages, or peeling. The patient’s axial temperature was 38.3°C. Upon auscultation of the
lungs, vesicular breathing was heard with no wheezes, and the
respiratory rate was 22 breaths per minute. The patient’s heart
sounds were rhythmic and muffled, with a heart rate of 88 beats
per minute and a blood pressure of 133/74 mmHg. The abdomen
was tender upon palpation in the left lower quadrant, with positive peritoneal symptoms. Rigidity of the neck muscles was noted
at the level of three transverse fingers.
Laboratory data showed the following: A complete blood
count on the day of hospitalization revealed a red blood cell
count of 3.69×109
/l, hemoglobin level of 97 g/l, platelet count of
538×109
/l, leukocyte count of 26.5×109
/l, and the following differential: segmented neutrophils 80%, band neutrophils 16%, monocytes 2%, and lymphocytes 2%. A biochemical blood test on the
day of hospitalization showed the following results: ALT 81 U/l,
AST 72 U/l, total protein 66 g/l, albumin 31 U/l, amylase 40 g/l,
glucose 5.63 mmol/l, and C-reactive protein 272.7 mg/l.
Data from instrumental research methods included an ultrasound of the abdominal cavity, which showed signs of bilateral
calico ectasia and fluid in the pelvis with a volume of up to 300
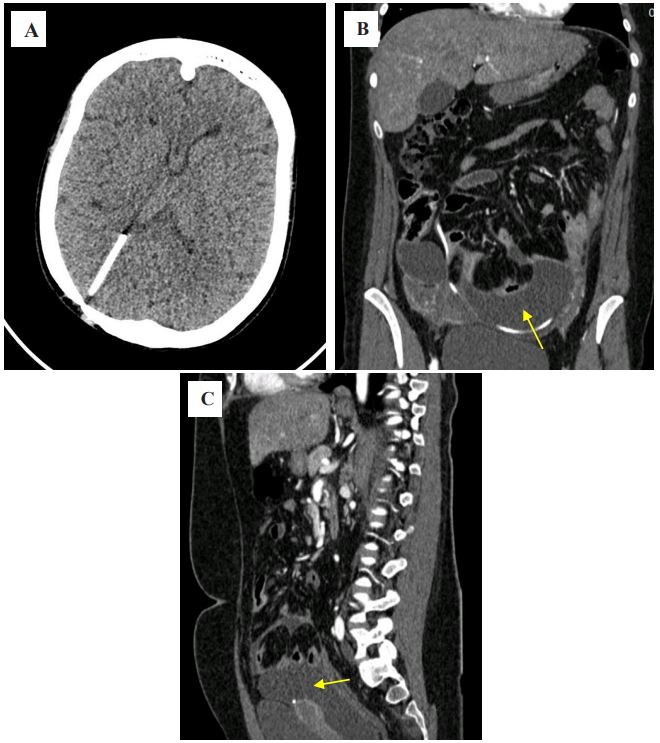
ml. A CT scan of the brain revealed a ventricular catheter in the
projection of the posterior horn of the right lateral ventricle (Figure 1A). A CT scan of the abdominal organs showed interloop
fluid in the lower floors of the abdominal cavity, in the projection
of the installed IPS, spreading into the pelvic cavity, with a layer
thickness of up to 4.5 cm, compressing the intestinal loops (Figure
1B,C).
The patient was examined by a general surgeon and, with
suspected peritonitis, was urgently taken to the operating room,
where diagnostic laparoscopy was performed. Turbid white fluid
was detected in the abdominal cavity and the peritoneal end of
the VPS was visualized. Access conversion was then performed,
followed by laparotomy, externalization of the shunt, and drainage of the abdominal cavity. Samples of cerebrospinal fluid from
the shunt system and the contents of the abdominal cavity were
sent for laboratory and bacteriological examination.
The results of the analysis of the cerebrospinal fluid obtained
from the shunt system showed a protein level of 1.1 g/l and glucose level of 3.16 g/l. Cell count was found to be 11008×106
/l,
with 92% neutrophils and 8% lymphocytes. A microbiological (bacteriological) study of the cerebrospinal fluid revealed the
growth of Streptococcus suis. Similarly, a bacteriological study of
the exudate from the abdominal cavity also showed the growth of
Streptococcus suis. An antibiogram revealed the sensitivity of the
microorganism to a wide range of antibiotics, including Benzylpenicillin, Vancomycin, Clindamycin, Cefotaxime, Erythromycin,
and Levofloxacin.
Based on clinical, laboratory, instrumental, and intraoperative
data, the patient was diagnosed with primary bacterial meningitis caused by Streptococcus suis, classified as moderate to severe
and complicated by secondary shunt-associated diffuse fibrinous peritonitis. In the postoperative period, the patient received
treatment in the neurosurgical department, including antibacterial therapy with Ceftriaxone (4 g per day), Vancomycin (2 g per
day), Ciprofloxacin (1 g per day), and symptomatic therapy. The
patient’s condition showed positive progress, with a regression of
infectious-inflammatory and pain symptoms and an improvement
in overall well-being.
Laboratory parameters were closely monitored, and additional
cerebrospinal fluid samples were taken from the distal end of the
functioning peritoneal shunt for laboratory and bacteriological
examination.
On the 27th day of illness, the patient’s complete blood count
showed the following results: Red blood cells 3.62×109
/l, hemoglobin 97 g/l, platelets 538×109
/l, leukocytes 3.62×109
/l, segmented neutrophils 72%, band neutrophils 5%, monocytes 4%,
and lymphocytes 13%. A biochemical blood test on the same day
revealed the following values: ALT 25 U/l, AST 31 U/l, total protein
81 g/l, albumin 44 U/l, amylase 90 g/l, glucose 4.8 mmol/l, and
C-reactive protein 23.9 mg/l. The results of cerebrospinal fluid
analysis on the 27th day of illness showed a protein level of 0.33
g/l, glucose level of 3.74 g/l, and cell count of 1×106
/l, with 32%
neutrophils and 68% lymphocytes. A bacteriological study did not
reveal the growth of any aerobic or anaerobic microorganisms.
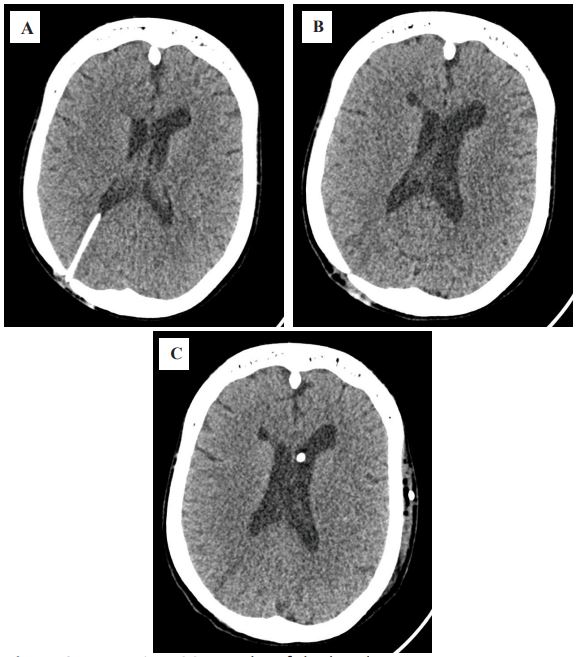
During the follow-up MSCT, normalization of the ventricular size
was observed (Figure 2), with no signs of increasing hydrocephalus
On the 28th day after the onset of the disease, an operation
was performed to remove the externalized ventriculoperitoneal
shunt and install a new one. This was done after the infectiousinflammatory syndrome had completely regressed and laboratory
parameters had normalized. The new shunt was placed from the
left Kocher point to the left flank region of the abdominal cavity.
The postoperative period went smoothly without any complications. The wounds healed well, and the sutures were removed on
the 10th day. The patient was discharged in satisfactory condition
on the 37th day for outpatient treatment.
Discussion
Streptococcus suis (S. suis) is a Gram-positive facultative anaerobe and a major pathogen of pigs that can be transmitted to humans [13]. Meningitis is the most common disease caused by this
microorganism in humans, with the greatest danger being the development of sepsis and a high risk of death. Other clinical manifestations include enteritis, endocarditis, arthritis, endophthalmitis, uveitis, and spondylodiscitis [14-16]. Permanent hearing loss
or vestibular dysfunction are the most common consequences of
S. suis infection, especially in patients with meningitis. However,
the most dangerous complication is the development of sepsis
with a high risk of death [14]. Since the first reported case of human infection with S. suis in Denmark in 1968, more than 700
cases have been reported worldwide [17]. The majority of reported cases of human infection with S. suis occur in Southeast Asian
countries, which are characterized by high population densities,
large numbers of pig farms, and a national cuisine that involves
eating raw or lightly cooked pork [16]. According to the literature,
S. suis is susceptible to penicillin antibiotics, Ceftriaxone, and Vancomycin [18]. In our case, the patient was treated with Ceftriaxone, Ciprofloxacin, and Vancomycin.
Conclusion
The development of VPS-associated peritonitis as a serious
complication of meningitis highlights the need for improved anti-epidemic measures for those working with pigs or consuming
pork products. The incidence of diseases caused by S. suis, a new
pathogen for humans, is increasing.
Declarations
Institutional review board statement: The work was carried
out according to the principles of voluntariness and confidentiality in accordance with Federal Law “On the Basics of Health Protection of Citizens in Russian Federation” 21.11.2011 N 323-FZ,
and the Helsinki Declaration on Human Rights.
Funding: This research was conducted without sponsorship.
Conflicts of interest: The authors declare no conflicts of interest.
References
- Samochernykh NK, Abramov KB, Nikolaenko MS, Sakhno LV, Samochernykh KA, et al. The treatment of patients with posthemorrhagic hydrocephalus. Rossiyskiy Vestnik Perinatologii i Pediatrii. 2021; 66 (5): 97-104.
- Wong T, Gold J, Houser R, Herschman Y, Jani R, Goldstein I. Ventriculopleural shunt: Review of literature and novel ways to improve ventriculopleural shunt tolerance. J Neurol Sci. 2021; 428: 117564. doi: 10.1016/j.jns.2021.117564.
- Reddy GK, Bollam P, Caldito, G. Long term outcomes of ventriculoperitoneal shunt surgery in patients with hydrocephalus. World Neurosurg. 2014; 404-410.
- Di Rocco C, Marchese E, Velardi F. A survey of the first complications of newly implanted CSf shunt devices for the treatment of nontumoral hydrocephalus. Child’s Nerv. Syst. 1994; 10: 321-327. doi:10.1007/Bf00335171.
- Christoph CL, Poole CA, Kochan PS. Operative gastric perforation: A rare complication of ventriculoperitoneal shunt. Pediatr Radiol. 1995; 25(1): S173-4.
- Van Dong H, Van HD, Vu HT, Chu HT. Duodenal perforation as a postoperative complication after ventriculoperitoneal shunt: A case report. Int J Surg Case Rep. 2021; 83: 106059.
- Alonso-Vanegas M, Alvarez JL, Delgado L, Mendizabal R, Jimenez JL, et al. Gastric perforation due to ventriculo-peritoneal shunt. Pediatr Neurosurg. 1994; 21: 192-4.
- Martinez Hernandez-Magro P, Barrera Roman C, Villanueva Saenz E, Zavala MJ. Colonic perforation as a complication of ventriculoperitoneal shunt: A case report. Tech Coloproctol. 2006; 10: 353-5.
- Mukhida K, Sharma MR, Shilpakar SK. Management of hydrocephalus with ventriculoperitoneal shunts: Review of 274 cases. Nepal Journal of Neuroscience. 2004; 1: 104-112.
- Lai KK. Enterobacter sakazakii infections among neonates, infants, children, and adults. Case reports and a review of the literature. Medicine (Baltimore). 2001; 80(2): 113-22. doi: 10.1097/00005792-200103000-00004.
- Mizuno S, Koyama J, Kurosawa H, Kasai M. Treatment optimization by monitoring vancomycin concentration in the serum and cerebrospinal fluid in a child with cystoperitoneal shunt-related infection caused by methicillin-resistant Staphylococcus aureus: A case report and literature review. Childs Nerv Syst. 2023; 39(11): 3307-3310. doi: 10.1007/s00381-023-06004-0.
- Hanak BW, Bonow RH, Harris CA, Browd SR. Cerebrospinal Fluid Shunting Complications in Children. Pediatr Neurosurg. 2017; 52(6): 381-400. doi: 10.1159/000452840.
- Perch B, Kristjansen P, Skadhauge K. Group R streptococci pathogenic for man. Two cases of meningitis and one fatal case of sepsis. Acta Pathol Microbiol Scand. 1968; 74: 69-76.
- Wertheim HF, Nghia HD, Taylor W, Schultsz C. Streptococcus suis: An emerging human pathogen. Clin Infect Dis. 2009; 48: 617-625.
- Suankratay C, Intalapaporn P, Nunthapisud P, Arunyingmongkol K, Wilde H. Streptococcus suis meningitis in Thailand. Southeast Asian J Trop Med Public Health. 2004; 35: 868-876.
- Choi SM, Cho BH, Choi KH, Nam TS, Kim JT, et al. Meningitis caused by Streptococcus suis: Case report and review of the literature. J Clin Neurol. 2012; 8(1): 79-82. doi: 10.3988/jcn.2012.8.1.79.
- Huang YT, Teng LJ, Ho SW, Hsueh PR. Streptococcus suis infection. J Microbiol Immunol Infect. 2005; 38: 306-313.
- Gottschalk M, Segura M, Xu J. Streptococcus suis infections in humans: The Chinese experience and the situation in North America. Anim Health Res Rev. 2007; 8: 29-45.